��Ŀ����
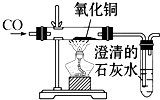
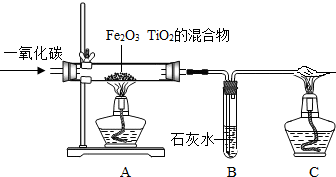
 ��ͼΪһ����̼��ԭ����ͭ��װ��ͼ��
��ͼΪһ����̼��ԭ����ͭ��װ��ͼ����1��ͼ��A����ʵ��������
��ɫ������Ϊ��ɫ
��ɫ������Ϊ��ɫ
����ѧ����ʽΪCO+CuO
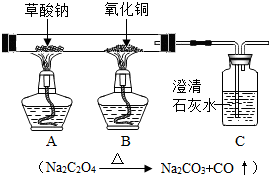
Cu+CO2
| ||
CO+CuO
Cu+CO2
��B����ʵ��������
| ||
�����ʯ��ˮ�����
�����ʯ��ˮ�����
����ѧ����ʽΪCO2+Ca��OH��2=CaCO3��+H2O
CO2+Ca��OH��2=CaCO3��+H2O
����2����װ�ô��ڵ���Ҫ������
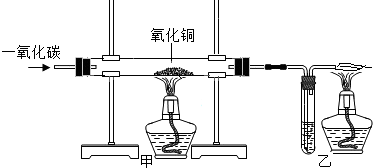
�ӵ������ų���β���к���һ����̼����Ⱦ����
�ӵ������ų���β���к���һ����̼����Ⱦ����
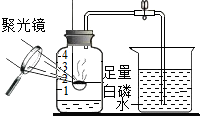
���Ľ��ľ����ʩ����β����ȼ��β�������ռ����������ɣ�
��β����ȼ��β�������ռ����������ɣ�
����������1��һ����̼�л�ԭ�ԣ��ɽ���ɫ������ͭ��ԭΪ��ɫ��ͭ��ͬʱ���ɶ�����̼��������̼��ʹ�����ʯ��ˮ����ǣ�����Ϸ�Ӧԭ����д����ʽ��
��2��һ����̼���ж��ԣ��ŷŵ������л���Ⱦ�������ɽ����ռ������������������ȼ�Խ���ȼ�յ���
��2��һ����̼���ж��ԣ��ŷŵ������л���Ⱦ�������ɽ����ռ������������������ȼ�Խ���ȼ�յ���
����⣺��1��һ����̼�л�ԭ�ԣ��ɽ���ɫ������ͭ��ԭΪ��ɫ��ͭ��ͬʱ���ɶ�����̼��������̼��ʹ�����ʯ��ˮ����ǣ�����ʽ�ֱ���CO+CuO
Cu+CO2��CO2+Ca��OH��2=CaCO3��+H2O��
��2��һ����̼�ж����ŷŵ������л���Ⱦ���������Բ���ֱ�Ӱ�β���ŵ������У�Ӧ�õ�ȼʹ�����ɶ�����̼��β���ռ�������
�ʴ�Ϊ����1��ɫ������Ϊ��ɫ�� CO+CuO
Cu+CO2������ʯ��ˮ����ǣ� CO2+Ca��OH��2=CaCO3��+H2O��
��2���������ų���β���к���һ����̼����Ⱦ��������β����ȼ��β�������ռ����������ɣ���
| ||
��2��һ����̼�ж����ŷŵ������л���Ⱦ���������Բ���ֱ�Ӱ�β���ŵ������У�Ӧ�õ�ȼʹ�����ɶ�����̼��β���ռ�������
�ʴ�Ϊ����1��ɫ������Ϊ��ɫ�� CO+CuO
| ||
��2���������ų���β���к���һ����̼����Ⱦ��������β����ȼ��β�������ռ����������ɣ���
������������Ҫ������һ����̼��ԭ����ͭʵ�飬��ȷ��ʵ������ԭ����ʵ�鲽��ȣ�����Ҫע��һ����̼��β������������˳�����

��ϰ��ϵ�д�
�����Ŀ

 ����������벻����������
����������벻����������