��Ŀ����
8���ּס�������ѧС�鰲װ������ͼ1װ�ã�ͨ���ⶨ������ͬ�����������ʱ�䳤����̽��Ӱ��H2O2�ֽ����ʵ�������1�����з����ܼ���װ�������Ե���AB
A����ע���������̶������Һ©����ע����������ˮ����Һ©�������������Һ©���������γ��ȶ���ˮ���������������ã���֮�����Բ
B���رշ�Һ©��������������ƿ�����ע�����������ƣ����ƣ���ֹͣ���Ⱥ�ע���������ص�ԭ��λ�ã������������ã���֮�����Բ
C���رշ�Һ©��������������ע�����������ƣ����ƣ���ֹͣ������ע�����������ƶ��������������ã���֮�����Բ
��2��MnO2������H2O2�ֽ�Ļ�ѧ����ʽ��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2����
��3����С��������ʵ����Ʒ�������������
| ʵ���� | ʵ��Ŀ�� | �¶� | ���� | Ũ�� |
| ����ʵ��� | ̽��Ũ�ȶ�H2O2�ֽ����ʵ�Ӱ�� | 25�� | �������� | 10mL��2%H2O2 |
| ����ʵ��� | 25�� | �������� | 10mL��5%H2O2 |
����ʵ��ó������ݿ�֪��Ũ��Խ��H2O2�ֽ�����Խ�죻
�������о����ᡢ���H2O2�ֽ�Ӱ�����ص����ݷ�������ͬ�����£�H2O2�ڼ��ᡱ����Ի����·ų��������ʽϿ죻�������������BaO2������������Һ��Ӧ��H2O2����֪���������ᱵ���������仯ѧ��Ӧ����ʽΪBaO2+H2SO4=BaSO4��+H2O2
֧����һ�����������ǹ������������Ի����·ֽ����

���� ��1�������Լ����ԭ��������װ�ú�ˮ���ɷ�յ����壬�ı�ijһ����ʹѹǿ�����ı����ж������Եĺû���
��2��MnO2����H2O2�ֽ�Ϊ������ˮ��
��3������ʵ��Ҫ����ֻ����һ���������˱���ΪH2O2��Ũ�ȣ������Ӧ����һ�£�
��4���������-ʱ��ͼ��б�ʼ��ɴ�����ѧ��Ӧ���ʣ�
���������о����ᡢ���H2O2�ֽ�Ӱ�����ص����ݷ�����֪����Խǿ���ų����������Խ�죮
��� �⣺��1�������Եļ����Ǹ���ѹǿԭ�������Թرշ�Һ©����������ע�������ƿ�ֹͣ������ע���������ܹ���ԭ��Ҳ�ɹ̶�ע��������������Һ����Ҳ���������¶ȷ�����ѡAB��
��2��H2O2��MnO2���·ֽ�����ˮ����������ѧ����ʽΪ��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$O2��+2H2O��
��3���ɼ�������ʵ��Աȿ��Կ���ʵ����д�����ʵ����е���ͬ������������Ũ�Ȳ�ͬ����˼����ʵ��Ŀ��Ϊ̽��Ũ�ȶ����ʵ�Ӱ�죻
��4������ͼ���֪�����-ʱ��ͼ��б�ʼ��ɴ�����ѧ��Ӧ���ʣ��ɼ���ʵ���������߿�֪������ʵ���б�ʴ����H2O2��Ũ��Խ��ֽ�����Խ�죬
���������о����ᡢ���H2O2�ֽ�Ӱ�����ص����ݷ�����֪����Խǿ���ų����������Խ�죬������֪BaO2������H2SO4��Һ��Ӧ��H2O2�ķ�Ӧ��ΪBaO2��H2SO4������ΪH2O2������ԭ���غ��֪��һ�ֲ���Ϊ���ᱵ������BaO2+H2SO4=H2O2+BaSO4������ͼ��֪���������£�H2O2�ֽ����������
�ʴ�Ϊ��
��1��AB����2��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2������3��Ũ�ȣ�
��4����Խ�죻�ڼBaO2+H2SO4=BaSO4��+H2O2���������������Ի����·ֽ������
���� ���⿼��������ضԻ�ѧ��Ӧ���ʵ�Ӱ�죬�漰�����Եļ��鷽����Ҫ����������𰸲�Ψһ����ԭ��������װ�ú�ˮ���ɷ�յ����壬�ı�ijһ����ʹѹǿ�����ı����ж������Եĺû��������ۺ��Խ�ǿ���Ѷ����У�

| A�� | ����2.3% | B�� | ����4% | C�� | ����2.3%��4% | D�� | ����4% |
| A�� | 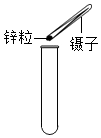 ȡ��п�� | B�� |  ����Һ�� | ||
| C�� | 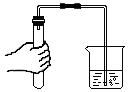 ���װ�������� | D�� | 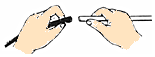 ������������Ƥ�� |
| A | B | C | D | ||
| ת����ϵ |  |  |  |  | |
| ���� | �� | C | Na2CO3 | CO2 | Fe |
| �� | O2 | Ca��OH��2 | CaCO3 | HCl | |
| �� | CuO | MgCl2 | NaOH | CuCl2 | |
| �� | CO2 | ϡH2SO4 | Na2CO3 | NaOH | |
| A�� | A | B�� | B | C�� | C | D�� | D |
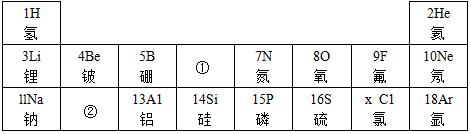
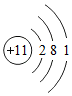 ����Ԫ�ص�ԭ�ӽṹʾ��ͼ����ԭ���ڻ�ѧ��Ӧ����ʧȥ���ӣ���õ�����ʧȥ�������ڻ�ѧ��Ӧ���γɵ�������Ne����Ԫ�ط��ţ�ԭ�Ӿ�����ͬ�ĺ�������Ų���
����Ԫ�ص�ԭ�ӽṹʾ��ͼ����ԭ���ڻ�ѧ��Ӧ����ʧȥ���ӣ���õ�����ʧȥ�������ڻ�ѧ��Ӧ���γɵ�������Ne����Ԫ�ط��ţ�ԭ�Ӿ�����ͬ�ĺ�������Ų���