��Ŀ����
6�� ��ѧ�о�С��Ϊ�ⶨijʯ��ʯ��Ʒ��̼��Ƶ�����������ȡ25gʯ��ʯ��Ʒ�����ձ��У�Ȼ����������μ���һ����ij����������ϡ���ᣬʹ֮����Ʒ��ַ�Ӧ�����ʲ��μӷ�Ӧ�������ŷ�Ӧ���У�����ϡ����������뷴Ӧ�õ�����������仯��ϵ��ͼ��ʾ����������м������ݣ�
��ѧ�о�С��Ϊ�ⶨijʯ��ʯ��Ʒ��̼��Ƶ�����������ȡ25gʯ��ʯ��Ʒ�����ձ��У�Ȼ����������μ���һ����ij����������ϡ���ᣬʹ֮����Ʒ��ַ�Ӧ�����ʲ��μӷ�Ӧ�������ŷ�Ӧ���У�����ϡ����������뷴Ӧ�õ�����������仯��ϵ��ͼ��ʾ����������м������ݣ���1����Ʒ��̼��Ƶ����������Ƕ��٣�
��2������ϡ�������ʵ����������Ƕ��٣�
��3������146gϡ���ᷴӦ��������Һ���������������Ƕ��٣���������0.1%��
���� ̼��ƺ�ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼�������ṩ�����ݺͷ�Ӧ�Ļ�ѧ����ʽ���Խ�����ط���ļ�����жϣ�
��� �⣺��1����25g��Ʒ��̼�������Ϊx������ϡ���������ʵ�����Ϊy����Ӧ�������Ȼ�������Ϊz��
CaCO3+2HCl=CaCl2+H2O+CO2����
100 73 111 44
x y z 8.8g
$\frac{100}{x}$=$\frac{73}{y}$=$\frac{111}{z}$=$\frac{44}{8.8g}$��
x=20g��y=14.6g��z=22.2g��
��Ʒ��̼��Ƶ����������ǣ�$\frac{20g}{25g}$��100%=80%��
����Ʒ��̼��Ƶ�����������80%��
��2������ϡ�������ʵ����������ǣ�$\frac{14.6g}{146g}$��100%=10%��
������ϡ�������ʵ�����������10%��
��3������146gϡ���ᷴӦ��������Һ���������������ǣ�$\frac{22.2g}{20g+146g-8.8g}$��100%=14.1%��
�𣺼���146gϡ���ᷴӦ��������Һ����������������14.1%��
���� ������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ�����������ʱҪע��淶�Ժ�ȷ�ԣ�

��ϰ��ϵ�д�
�����Ŀ
1����һ���������ۼ��뵽AgNO3��Cu��NO3��2�Ļ����Һ�У�������AgNO3��Cu��NO3��2ǡ����ȫ��Ӧ���ˣ������µķ�������ȷ���ǣ�������
| A�� | ��ֽ�����µ�������ͭ | B�� | ��Һ�е�������Fe��NO3��2��AgNO3 | ||
| C�� | ��Һ�е�������AgNO3 | D�� | ��ֽ�����µ�������ͭ���� |
15�����Ʊ�������ʵ��̽������Ҫ������������ͼʵ�鷽��������У��ܴﵽʵ��Ŀ���ǣ�������
| A�� |  �Ƚ�����ͭ�����ᷴӦ�ľ��ҳ̶� | |
| B�� |  ̽����������ʴ�Ƿ���Ҫˮ | |
| C�� |  ̽��MnO2��H2O2�ֽ�Ĵ����� | |
| D�� |  ̽����ȼ���ȼ���Ƿ���Ҫ�ﵽһ���¶� |
16�����ջ�ѧ������ѧ�û�ѧ�Ĺؼ������л�ѧ����������������������ϵ��ǣ�������
| A�� | O2-2����ԭ�� | B�� | 2H2O-2��ˮ���� | C�� | 3H-3����Ԫ�� | D�� | Na-������ |
 ��
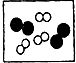
�� �ֱ��ʾ���ֲ�ͬԪ�ص�ԭ�ӣ����б�ʾ��������ǣ�������
�ֱ��ʾ���ֲ�ͬԪ�ص�ԭ�ӣ����б�ʾ��������ǣ�������