��Ŀ����
��ʯ����ʵ���ҳ��õĸ������
[�������]ʵ�����о��õļ�ʯ����Ʒ�ijɷ���ʲô��
[���в���](1)���ü�ʯ����Ʒ�п��ܺ���CaO��NaOH��Ca(OH)2��Na2CO3��CaCO3�ȳɷ֡��������������У����ڼ����____����Ʒ�п��ܺ���Ca(OH)2��Na2CO3��ԭ����______________________��________________________�����û�ѧ����ʽ��ʾ��
[ʵ�����]
[�������]ʵ�����о��õļ�ʯ����Ʒ�ijɷ���ʲô��
[���в���](1)���ü�ʯ����Ʒ�п��ܺ���CaO��NaOH��Ca(OH)2��Na2CO3��CaCO3�ȳɷ֡��������������У����ڼ����____����Ʒ�п��ܺ���Ca(OH)2��Na2CO3��ԭ����______________________��________________________�����û�ѧ����ʽ��ʾ��
[ʵ�����]

�Իش��������⣺
(2)�����ٵ�������____��
(3)����ʵ���������������˵����ȷ����____������ţ���
A����Ʒ��ˮ�������ڷ��ȣ�˵����Ʒ��һ������CaO
B����Ʒ������ˮ�����й��壬˵����Ʒ��һ������CaCO3
C����ҺA����CaCl2��Һ��������壬˵����Ʒ��һ������Na2CO3
D����ҺC�����̪��죬˵����Ʒ��һ������NaOH
(4)����ҺA�м���CaCl2��Һ���жϼ���CaCl2��Һ�����ķ�����_____________________��
(5)д����ҺC�����������ӵķ���____��
[ʵ�����]�þ��ü�ʯ����Ʒ�п϶����еijɷ���____���ѧʽ���������ɷ��в���ȷ����
(2)�����ٵ�������____��
(3)����ʵ���������������˵����ȷ����____������ţ���
A����Ʒ��ˮ�������ڷ��ȣ�˵����Ʒ��һ������CaO
B����Ʒ������ˮ�����й��壬˵����Ʒ��һ������CaCO3
C����ҺA����CaCl2��Һ��������壬˵����Ʒ��һ������Na2CO3
D����ҺC�����̪��죬˵����Ʒ��һ������NaOH
(4)����ҺA�м���CaCl2��Һ���жϼ���CaCl2��Һ�����ķ�����_____________________��
(5)д����ҺC�����������ӵķ���____��
[ʵ�����]�þ��ü�ʯ����Ʒ�п϶����еijɷ���____���ѧʽ���������ɷ��в���ȷ����
(1)NaOH��Ca(OH)2��CaO+H2O==Ca(OH)2��2NaOH+CO2==Na2CO3+H2O
(2)����
(3)C
(4)ȡ�����ϲ���Һ���μ�CaCl2��Һ�����ް�ɫ������������˵��������Լ�����
(5)OH-��Cl-��Na2CO3
(2)����
(3)C
(4)ȡ�����ϲ���Һ���μ�CaCl2��Һ�����ް�ɫ������������˵��������Լ�����
(5)OH-��Cl-��Na2CO3

��ϰ��ϵ�д�
�����Ŀ
 33����ʯ����ʵ���ҳ��õĸ������
33����ʯ����ʵ���ҳ��õĸ������
 ��ʯ����ʵ���ҳ��õĸ������ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽����
��ʯ����ʵ���ҳ��õĸ������ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽���� 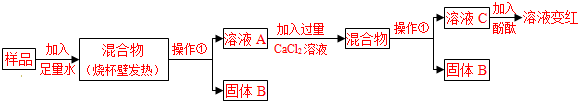

 �÷�Ӧ���� ��10�� ����д������Ӧ���ͣ��� ���������غ㶨�ɣ�������֪�����С�����������Ļ�ѧʽ�� ��11�� ��
�÷�Ӧ���� ��10�� ����д������Ӧ���ͣ��� ���������غ㶨�ɣ�������֪�����С�����������Ļ�ѧʽ�� ��11�� ��
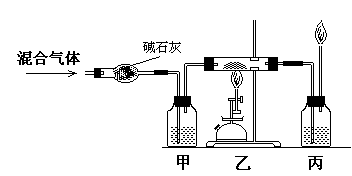
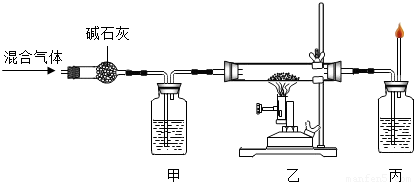
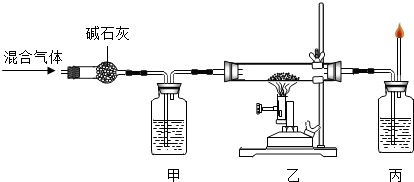
 ��17�� ���÷�Ӧ�л�ԭ���� ��18�� �� �ס���������ʢ�ŵ���Һ��
��17�� ���÷�Ӧ�л�ԭ���� ��18�� �� �ס���������ʢ�ŵ���Һ��