��Ŀ����
��2011?��ƽ����ģ����һ�ձ���ʢ��20.4g Na2CO3��NaCl ��ɵĹ�������������μ�������������Ϊ10%��ϡ���ᣮ�ų��������������������ϡ�����������ϵ������ͼ��ʾ�����������ش����⣺��1�����μ�ϡ������ͼ��B��ʱ���ձ�����Һ��pH______7�����=��������
��2��ԭ�������̼���Ƶ�������
��3�����μ�ϡ������ͼ��A��ʱ���ձ���Ϊ��������Һ�����£���ͨ����������������ʵ�����������������������һλС������

���𰸡�����������ͼ���ѿ��������μ�ϡ������ͼ��A��ʱ�����ɵ�����������������ӣ�˵��̼�����Ѹպ���ȫ��Ӧ�����ټ�ϡ��������������ѹ�������˵���μӷ�Ӧ��ϡ���������73g����ϡ���������ʵ��������뻯ѧ����ʽ���м���Ϳ�����йص�������Ӧ�����õ��������Ը��ݼ������ʵ�������-������ˮҲ���μӷ�Ӧ�����ʵ�����-���ɵ�����������������
����⣺��1����ͼ���в��ѿ��������μ�ϡ���ᵽAʱ��������̼�������������ӣ�˵��̼��������ȫ��Ӧ�����ٵμ�ϡ���ᣬ�������������Һ�����ԣ�PHС��7�������
��2����3����ϡ��������������ݻ�ѧ����ʽ���м��㣻
�ʴ�Ϊ��73g10%��ϡ�����к�HCl�������ǣ�73g×10%=7.3g����μӷ�Ӧ��̼���Ƶ�����Ϊx����Ӧ���ɵ��Ȼ��Ƶ�����Ϊy�����ɵĶ�����̼������Ϊz��
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 73 117 44
x 7.3g y z
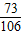 =
=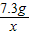 ��x=10.6g
��x=10.6g
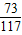 =
=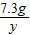 ��y=11.7g
��y=11.7g
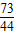 =
=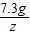 ��z=4.4g
��z=4.4g
ԭ�������̼���Ƶ�����Ϊ10.6g��
���ﲻ������Һ�����ʵ�����Ϊ��11.7g+��20.4g-10.6g��=21.5g
�ձ��ﲻ������Һ������Ϊ��20.4g+73g-4.4g=89g
�ձ��ﲻ������Һ�����ʵ���������Ϊ��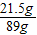 ×100%=24.2%
×100%=24.2%
�����ʵ���������Ϊ24.2%��
�����������ѶȽϴ���Ҫ�����˸��ݻ�ѧ����ʽ���м�����й���Һ���������������ļ��㣬�Ӷ�����ѧ���Ը����Ӧ�úͷ������⡢��������������
����⣺��1����ͼ���в��ѿ��������μ�ϡ���ᵽAʱ��������̼�������������ӣ�˵��̼��������ȫ��Ӧ�����ٵμ�ϡ���ᣬ�������������Һ�����ԣ�PHС��7�������
��2����3����ϡ��������������ݻ�ѧ����ʽ���м��㣻
�ʴ�Ϊ��73g10%��ϡ�����к�HCl�������ǣ�73g×10%=7.3g����μӷ�Ӧ��̼���Ƶ�����Ϊx����Ӧ���ɵ��Ȼ��Ƶ�����Ϊy�����ɵĶ�����̼������Ϊz��
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 73 117 44
x 7.3g y z
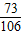 =
=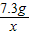 ��x=10.6g
��x=10.6g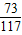 =
=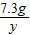 ��y=11.7g
��y=11.7g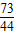 =
=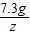 ��z=4.4g
��z=4.4gԭ�������̼���Ƶ�����Ϊ10.6g��
���ﲻ������Һ�����ʵ�����Ϊ��11.7g+��20.4g-10.6g��=21.5g
�ձ��ﲻ������Һ������Ϊ��20.4g+73g-4.4g=89g
�ձ��ﲻ������Һ�����ʵ���������Ϊ��
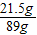 ×100%=24.2%
×100%=24.2%�����ʵ���������Ϊ24.2%��
�����������ѶȽϴ���Ҫ�����˸��ݻ�ѧ����ʽ���м�����й���Һ���������������ļ��㣬�Ӷ�����ѧ���Ը����Ӧ�úͷ������⡢��������������

��ϰ��ϵ�д�
�����Ŀ
��2011?��ƽ����ģ��ʵ������5�ְ�ɫ���壬��ʦ�û�ѧʵ��С��ͬѧ������Щ���壮
��1�������о�
��5�ְ�ɫ����ֱ��ǣ�NaOH��Na2CO3��CuSO4��Ca��OH��2��NaHCO3��
�ڲ������ϣ�С��ͬѧ������NaOH��Na2CO3��NaHCO3��һЩ���ʣ�
��2��ʵ��̽����ͬѧ�ǵ�̽���������£����������ɣ�
��3��ʵ�鷴˼����ʦ����ͬѧ������������ʵ��̽�����̣�����ȫ�淴˼������ʵ��������в�ȷ���ԣ�����ȷ����������______���ѧʽ������������������������
��1�������о�
��5�ְ�ɫ����ֱ��ǣ�NaOH��Na2CO3��CuSO4��Ca��OH��2��NaHCO3��
�ڲ������ϣ�С��ͬѧ������NaOH��Na2CO3��NaHCO3��һЩ���ʣ�
| ���� | NaOH | Na2CO3 | NaHCO3 |
| �ܽ��� | 20��ʱ�ܽ��109g | 20��ʱ�ܽ��21.5g �ܽ�ʱ���� | 20��ʱ�ܽ��9.6g �ܽ�ʱ���� |
| ���� | ���ֽ� | ���ֽ� | �����ֽⷴӦ���ֽ����Ϊ̼���ơ�������̼��ˮ�� |
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| ��һ������5֧�Թ��зֱ����������5�ְ�ɫ���壬 �ֱ����A��B��C��D��E��Ȼ�����μ���һ������ˮ���� | A��C��D����ҺΪ��ɫ��B����ҺΪ���ǣ�E����Һ����ɫ�� | B��______ E��______ | |
| �ڶ�����������A��C��D��Һ�зֱ�μ���ɫ�ķ�̪��Һ�� | ��֧�Թ��еķ�̪��Һ����죮 | ���ۣ� ______ | |
| ��������������ͼװ�ã���A��B��C����ʵ�飮 |  | ||
| �ټ��ȹ���A | B��Һ���������� | A��Na2CO3 | |
| �ڴ���ֹͣ���Ⱥ�ͨ������©����C��Һ�м���ϡ���� | C��Һ�������ݲ�����B��Һ���������ݳ����а�ɫ�������ɣ� | B��Һ�з�����Ӧ�Ļ�ѧ����ʽΪ______ | |
| ���IJ���������ͼװ�ã���B��C����ʵ�飮 |  | �����ȵ��Թܿ�����ɫҺ�����ɣ�B��Һ���������ݳ�����Һ����ǣ� | C��______ �����ų����ó��� D��NaOH |
| ʵ�鲽�� | ʵ������ͽ��� |
| ______ | ______ |


