��Ŀ����
���Ȼ��ƺ�̼�����(NH4HCO3)���Ʊ�̼�����ƺ��Ȼ��(NH4Cl)���÷�Ӧ
 �ɱ�ʾΪ��NaCl + NH4HCO3 NaHCO3 + NH4Cl��
�ɱ�ʾΪ��NaCl + NH4HCO3 NaHCO3 + NH4Cl��
20 ��ʱ����������ѧ����ʽ�з�Ӧ��������ȣ���100 gˮ�м���11.7 g NaCl��
15.8 g NH4HCO3������㣺
(1) �����ϴ���Һ���������������Ϊ����g��
(2) ��Ӧ��������Һ������NH4Cl�����������Ƕ��٣���д������ʽ���ɣ���
���ϣ�20 ��ʱ�������ʵ��ܽ�����£���������ͬʱ�ܽ���ˮ�и��Ե��ܽ�Ȳ��䡣
| ���� | NaCl | NH4HCO3 | NH4Cl | NaHCO3 |
| �ܽ��/g | 36.0 | 21.6 | 37.2 | 9.6 |
���⡿������NaHCO3������Ϊx��NH4Cl������Ϊy
NaCl + NH4HCO3 NaHCO3 + NH4Cl
58.5 84 53.5
11.7 x y
x =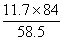 = 16.8g
= 16.8g
y =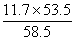 = 10.7g -------------------1��
= 10.7g -------------------1��
�����ϴ���Һ������NaHCO3���������=16.8-9.6=7.2g -------------------1��
��Һ������NH4Cl����������=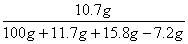 ��100% -----------------1��
��100% -----------------1��
�������ϴ���Һ������NaHCO3���������Ϊ7.2g����Ӧ��������Һ������NH4Cl��
��������Ϊ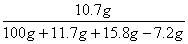 ��100%�������������������֣�
��100%�������������������֣�


 ��2012?����������Ϊ�����ṩ�˱������Ȼ��Դ��
��2012?����������Ϊ�����ṩ�˱������Ȼ��Դ��