��Ŀ����
���Ȼ��ƺ�̼����泥�NH4HCO3�����Ʊ�̼�����ƺ��Ȼ�泥��÷�Ӧ�ɱ�ʾΪ��NaCl+NH4HCO3�TNaHCO3+NH4Cl��20��ʱ��������ѧ����ʽ�з�Ӧ��������ȣ���100gˮ�м���11.7gNaCl��15.8gNH4HCO3����
��1�������Ͽ��Ƶ�NH4Cl������Ϊ
��2���÷�Ӧ�Ļ�����Ӧ������
��3�������ϴ���Һ���������������Ϊ���٣�����д���ڣ�3����Ľ�����̣�
���ϣ�20��ʱ�������ʵ��ܽ�����£���������ͬʱ�ܽ���ˮ�и��Ե��ܽ�Ȳ��䣮
��1�������Ͽ��Ƶ�NH4Cl������Ϊ
10.7g
10.7g
����2���÷�Ӧ�Ļ�����Ӧ������
���ֽⷴӦ
���ֽⷴӦ
����3�������ϴ���Һ���������������Ϊ���٣�����д���ڣ�3����Ľ�����̣�
���ϣ�20��ʱ�������ʵ��ܽ�����£���������ͬʱ�ܽ���ˮ�и��Ե��ܽ�Ȳ��䣮
| ���� | NaCl | NH4HCO3 | NH4Cl | NaHCO3 |
| �ܽ��/g | 36.0 | 21.6 | 37.2 | 9.8 |
����������20��ʱ NaCl��NH4HCO3���ܽ�ȿ�֪��������ȫ�ܽ⣬�ɷ���ʽ������֮���������ϵ��֪NaCl��NH4HCO3ǡ����ȫ��Ӧ��������NaCl�������������ɵ�NaHCO3��NH4Cl����������ϸ��¶������ʵ��ܽ�ȷ���������������ʼ������������1����3���⣻���ݸ��ֽⷴӦ�Ķ�����2����
����⣺��1�����ݻ�ѧ����ʽ��������ϵ����֪����NaCl��NH4HCO3ǡ����ȫ��Ӧ�������ɵ�NaHCO3������Ϊx��NH4Cl������Ϊy
NaCl+NH4HCO3�TNaHCO3+NH4Cl
58.5 79 84 53.5
11.7g x y
=
x=16.8g
=
y=10.7g
��2���÷�Ӧ�����ֻ�����������ɷ������������ֻ�����ķ�Ӧ�����ֽⷴӦ��
��3���������е��ܽ�ȱ�����֪����20��ʱ100gˮ���ܹ��ܽ�̼�����Ƶ�����Ϊ9.8g���ܹ��ܽ��Ȼ�淋�����Ϊ37.2g�����������ϴ���Һ��������NaHCO3���������Ϊ16.8g-9.8g=7.0g��
NaCl+NH4HCO3�TNaHCO3+NH4Cl
58.5 79 84 53.5
11.7g x y
| 58.5 |
| 11.7g |
| 84 |
| x |
x=16.8g
| 58.5 |
| 11.7g |
| 84 |
| x |
y=10.7g
��2���÷�Ӧ�����ֻ�����������ɷ������������ֻ�����ķ�Ӧ�����ֽⷴӦ��
��3���������е��ܽ�ȱ�����֪����20��ʱ100gˮ���ܹ��ܽ�̼�����Ƶ�����Ϊ9.8g���ܹ��ܽ��Ȼ�淋�����Ϊ37.2g�����������ϴ���Һ��������NaHCO3���������Ϊ16.8g-9.8g=7.0g��
���������⿼����ѧ�����������������龰��ͼ����Ϣ�������ѧ�ܽ�ȡ�����ʽ��������֪ʶ�ͼ��ܣ�ϸ�ĵ�̽����������������ĿҪ��������������������������

��ϰ��ϵ�д�
�����Ŀ
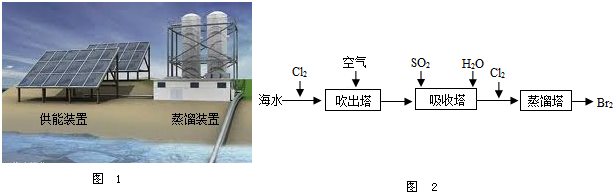
 ��2012?����������Ϊ�����ṩ�˱������Ȼ��Դ��
��2012?����������Ϊ�����ṩ�˱������Ȼ��Դ��