��Ŀ����
����ʹ�õ���ˮƿ�ײ���һ��ˮ����Ϊ�˳�ȥ��ˮƿ�ײ���ˮ�����о���ѧϰС���ͬѧ�����������о���ʵ�飺
���������ϡ�ˮ������Ҫ�ɷ���CaCO3��Mg(OH)2��Mg(OH)2�����ᷴӦ��ѧ����ʽΪMg(OH)2 + 2HCl =MgCl2 + 2H2O ��
��ʵ�顿ȡ50 gˮ������Ʒ�����ʲ��������ˮ��Ӧ�������ձ��У�������������������Ϊ10%���ᣬʹ��Ʒ��ȫ��Ӧ������¼��Ӧ�����л���������ı仯���ձ�����Ʒ��ϡ�����������m����ʼʱΪ300g��ʵ�����ݼ�¼���£�
���������ϡ�ˮ������Ҫ�ɷ���CaCO3��Mg(OH)2��Mg(OH)2�����ᷴӦ��ѧ����ʽΪMg(OH)2 + 2HCl =MgCl2 + 2H2O ��
��ʵ�顿ȡ50 gˮ������Ʒ�����ʲ��������ˮ��Ӧ�������ձ��У�������������������Ϊ10%���ᣬʹ��Ʒ��ȫ��Ӧ������¼��Ӧ�����л���������ı仯���ձ�����Ʒ��ϡ�����������m����ʼʱΪ300g��ʵ�����ݼ�¼���£�
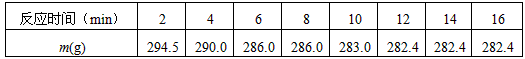
�����������
(1) �������м�¼�����ݣ�����Ϊ��Ӧʱ��Ϊ__________minʱ��¼�����������Ǵ���ġ�
(2) Mg(OH)2��þ������������Ԫ�ص�������Ϊ_______________________��
(3)��Ӧ��Ļ�����к���CaCl2 ��MgCl2��9.5 g MgCl2���е���Ԫ�ص�������_____________g HCl ���е���Ԫ�ص������൱��
(4)ˮ����̼��Ƶ����������Ƕ��٣�
(1) �������м�¼�����ݣ�����Ϊ��Ӧʱ��Ϊ__________minʱ��¼�����������Ǵ���ġ�
(2) Mg(OH)2��þ������������Ԫ�ص�������Ϊ_______________________��
(3)��Ӧ��Ļ�����к���CaCl2 ��MgCl2��9.5 g MgCl2���е���Ԫ�ص�������_____________g HCl ���е���Ԫ�ص������൱��
(4)ˮ����̼��Ƶ����������Ƕ��٣�
(1)8
(2) 12��16�� 1
(3)7.3
(4)��50 gˮ���к�CaCO3 ������Ϊx
CaCO3 + 2HCl ==CaCl2 + H2O + CO2 ��
100 44
x 17.6g

x= 40 g
ˮ����̼��Ƶ���������Ϊ=
(2) 12��16�� 1
(3)7.3
(4)��50 gˮ���к�CaCO3 ������Ϊx
CaCO3 + 2HCl ==CaCl2 + H2O + CO2 ��
100 44
x 17.6g

x= 40 g
ˮ����̼��Ƶ���������Ϊ=


��ϰ��ϵ�д�
 ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д�
�����Ŀ
����ʹ�õ���ˮƿ�ײ���һ��ˮ������Ҫ�ɷ���CaCO3��Mg��OH��2��ijѧУ��ѧ�о�С���ͬѧͨ��ʵ��ⶨˮ����CaCO3�ĺ�����ȡ200gˮ�������������ϡ���ᣬͬʱ����5���������������������������±���
�Իش��������⣺
��1��4���Ӻ�����CO2�������ӣ�˵��ˮ����CaCO3 �����ѻ�δ����Ӧ��ȫ��
��2������Ӧ��ȫʱ��CaCO3���ĵ��Ȼ���Ϊ g��
��3����ˮ����CaCO3�����������Ƕ��٣�
| ʱ��/min | 1 | 2 | 3 | 4 | 5 |
| ����/g | 30 | 50 | 60 | 66 | 66 |
��1��4���Ӻ�����CO2�������ӣ�˵��ˮ����CaCO3
��2������Ӧ��ȫʱ��CaCO3���ĵ��Ȼ���Ϊ
��3����ˮ����CaCO3�����������Ƕ��٣�
����ʹ�õ���ˮƿ�ײ���һ��ˮ������Ҫ�ɷ���̼��ƣ���ϡ����������ˮ����ijѧУ��ѧ������ȤС���ͬѧͨ��ʵ��ⶨˮ����̼��Ƶĺ�������������ϡ����ӵ�200gˮ���У�ͬʱ����4�����ڲ���CO2���������ֽ�����±���[CaCO3+2HCl�TCaCl2+H2O+CO2��]
��1����С�����û�м�¼��Ӧʱ��Ϊ180sʱ����
��M�����ɱ��������Ʋ⣬M= g��
��2����Ӧ����CO2��������Ϊ g����3������ʵ�����ݣ����ұߵ�����ֽ�ϻ�������CO2�����뷴Ӧʱ���ϵ�����ߣ������������Ʋ��ڷ�Ӧ60sʱ������CO2������ԼΪ ��
��4������ˮ����̼������������Ƕ��٣�
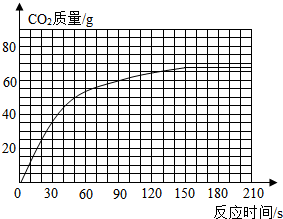
| ʱ��/s | 30 | 50 | 90 | 150 | 180 | 210 |
| CO2����/g | 30 | 50 | 60 | 66 | M | 66 |
��M�����ɱ��������Ʋ⣬M=
��2����Ӧ����CO2��������Ϊ
��4������ˮ����̼������������Ƕ��٣�

����ʹ�õ���ˮƿ�ײ���һ��ˮ������Ҫ�ɷ���CaCO3��Mg��OH��2��ijѧУ��ѧ�о�С���ͬѧͨ��ʵ��ⶨˮ����CaCO3�ĺ�����ȡ200gˮ�������������ϡ���ᣬͬʱ����5���������������������������±���
�Իش��������⣺
��1��4���Ӻ�����CO2�������ӣ�˵��ˮ����CaCO3 �����ѻ�δ����Ӧ��ȫ��
��2����ˮ����CaCO3�����������Ƕ��٣�
| ʱ��/min | 1 | 2 | 3 | 4 | 5 |
| ����/g | 30 | 50 | 60 | 66 | 66 |
��1��4���Ӻ�����CO2�������ӣ�˵��ˮ����CaCO3
��2����ˮ����CaCO3�����������Ƕ��٣�
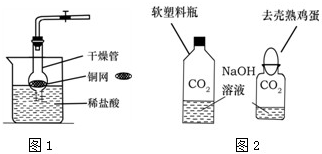 CO2�dz��л�ѧ�ص��о�������֮һ��ij��ѧ��ȤС�����������ʵ����̽��CO2����ȡ�����ʣ�
CO2�dz��л�ѧ�ص��о�������֮һ��ij��ѧ��ȤС�����������ʵ����̽��CO2����ȡ�����ʣ�