��Ŀ����
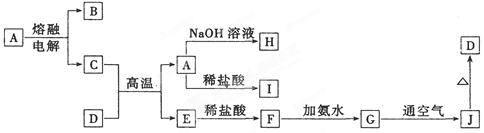
��10�֣����п�ͼ��ʾ������ת����ϵ�У������ճ������г����Ľ������ҡ��������dz��������嵥�ʡ�����B������C�������������İ�������E��A��ǿ�D�Ǻ�ˮ��Ũ����ߵ��Σ����ַ�Ӧ��������Pˮ����ȥ����

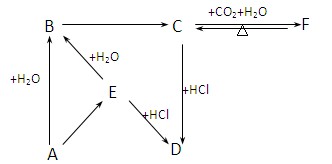
��ش��������⣺
��1��д�����������B�Ļ�ѧ����ʽ��
��2��д����A��Һ��Ӧ�����ӷ���ʽ��
��3������E��������ʱ��ȡ����E���Թ��� ��֤��E���и������ӡ�
��4��д��ʵ������ȡB�Ļ�ѧ����ʽ��
��5����ҵ����B��ԭ���Ʊ�ij�ᣬд�������Ũ��Һ�ڳ�������ij������Ӧ��������ɫ��Һ�Ļ�ѧ����ʽ��

��ش��������⣺
��1��д�����������B�Ļ�ѧ����ʽ��
��2��д����A��Һ��Ӧ�����ӷ���ʽ��
��3������E��������ʱ��ȡ����E���Թ��� ��֤��E���и������ӡ�
��4��д��ʵ������ȡB�Ļ�ѧ����ʽ��
��5����ҵ����B��ԭ���Ʊ�ij�ᣬд�������Ũ��Һ�ڳ�������ij������Ӧ��������ɫ��Һ�Ļ�ѧ����ʽ��
��10�֣���ÿ��2�֣���1��N2+3H2 2NH3
2NH3
��2��2Al+2OH-+2H2O=2AlO2-+3H2��
��3������NaOH��Һ�����ȣ���ʪ��ĺ�ɫʯ����ֽ�����Թܿ���ֽ����
��4��2NH4Cl+Ca(OH)2 CaCl2+2NH3��+2H2O
CaCl2+2NH3��+2H2O
��5��Cu+4HNO3(Ũ)=Cu(NO3)2+2NO2��+2H2O
 2NH3
2NH3��2��2Al+2OH-+2H2O=2AlO2-+3H2��
��3������NaOH��Һ�����ȣ���ʪ��ĺ�ɫʯ����ֽ�����Թܿ���ֽ����
��4��2NH4Cl+Ca(OH)2
 CaCl2+2NH3��+2H2O
CaCl2+2NH3��+2H2O��5��Cu+4HNO3(Ũ)=Cu(NO3)2+2NO2��+2H2O
����������ͼ�⣬������ͻ�Ƶ㡣����B������C�������������İ�������E������E���Ȼ�泥���C���Ȼ��⣬B�ǰ��������ǵ�������������������������D�Ǻ�ˮ��Ũ����ߵ��Σ�����D���Ȼ��ƣ���A���������ơ��������ܺ��������Ʒ�Ӧ�������������Լ�������

��ϰ��ϵ�д�
 ��ǰ�κ�ͬ����ϰϵ�д�
��ǰ�κ�ͬ����ϰϵ�д� ����С��ҵϵ�д�
����С��ҵϵ�д� �Ƹ�С״Ԫ����������ϰ��ϵ�д�
�Ƹ�С״Ԫ����������ϰ��ϵ�д� �ɹ�ѵ���ƻ�ϵ�д�
�ɹ�ѵ���ƻ�ϵ�д� ����ѵ����ֱͨ�п�����ϵ�д�
����ѵ����ֱͨ�п�����ϵ�д� һ���㶨ϵ�д�
һ���㶨ϵ�д� ��У��ҵ��ϵ�д�
��У��ҵ��ϵ�д�
�����Ŀ
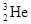 ������Ϊ�˾۱���ϣ����й���
������Ϊ�˾۱���ϣ����й��� ��Ϊͬλ��
��Ϊͬλ��