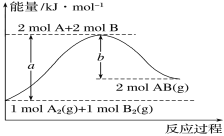
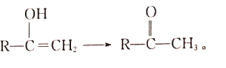
��Ŀ����
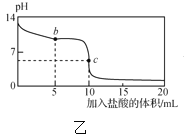
����Ŀ�������£���1.00mol��L-1�������20.00mL1.00mol��L-1�İ�ˮ�У���ҺpH���¶��������������ı仯������ͼ��ʾ��
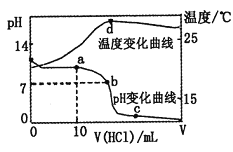
�����й�˵���в���ȷ���ǣ� ��
A. a����Һ������Ũ�ȴ�С��ϵ��c��NH4+����c��Cl-����c��OH-����c��H+��
B. b����Һ������Ũ�ȴ�С��ϵ��c��NH4+��=c��Cl-����c��H+��=c��OH-��
C. c����Һ������Ũ�ȴ�С��ϵ��c��NH4+��+c��H+��=c��Cl-��+c��OH-��
D. d��ʱ��Һ�¶ȴﵽ��ߣ�֮���¶������½���ԭ����NH3��H2O����
���𰸡�D
��������
A.a����Һ��Ϊ�����ʵ���Ũ�ȵ�![]() ��
��![]() ������Һ�Լ��ԣ��ʰ�ˮ�ĵ���̶ȴ���NH4Cl��ˮ��̶���������
������Һ�Լ��ԣ��ʰ�ˮ�ĵ���̶ȴ���NH4Cl��ˮ��̶���������![]() ��A����ȷ��
��A����ȷ��
B.b����Һ�����ԣ���![]() �����ݵ���غ���
�����ݵ���غ���![]() ������
������![]() ��B����ȷ��
��B����ȷ��
C.c����Һ��������NH4Cl��HCl�����ݵ���غ���![]() �� C����ȷ��
�� C����ȷ��
D.d��ʱ���ǡ���кͣ��ų�������࣬��Һ�¶ȴﵽ��ߣ��кͷ�Ӧ��������������¶ȵͣ���������ļ�������Һ�¶���������NH3��H2O�����أ�D�����ѡD��

��ϰ��ϵ�д�
�����Ŀ