��Ŀ����
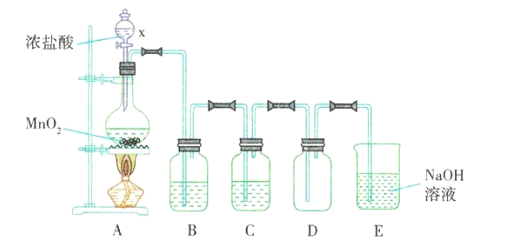
����Ŀ��ʵ���ҳ����������ƹ����70%��Ũ�����Ʊ�SO2���壬��Ӧ����ʽΪNa2SO3+H2SO4=Na2SO4+SO2��+H2O�������һ��ʵ��װ�ã��ռ�һ�Թܸ����SO2���塣
��1�������淽���У�A��ʾ�ɷ�Һ©������ƿ��ɵ����巢���������ڴ���ϵ�A����ɸ÷�Ӧ��ʵ��װ��ʾ��ͼ(�г�װ�á����ӽ��ܲ��ػ�����β���������ֱ��뻭������Ҫ���ȵ������·��������)��������������ÿ�������·������ĸB��C����������ѡ�õ�����(��������)���ױ�ʾ���£�___
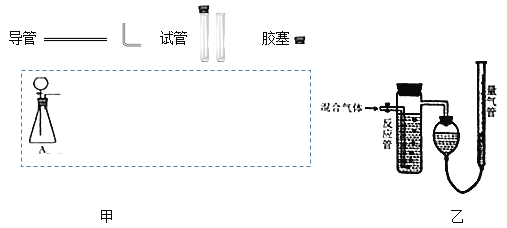
��2�����ݷ����е�װ��ͼ�ף��ڴ������д�±�(�ɲ�����)___
������� | �������������� | ���� |
A | �������ơ�Ũ���� | ����SO2 |
��3��ijʵ��С������ͼ��װ�òⶨ�ռ�����������SO2�ĺ�������Ӧ����װ�����Ը��������Һ��
��SO2�����Ը��������Һ������Ӧ�����ӷ���ʽΪ��___��
�ڷ�Ӧ������Һ�Ϻ�ɫ��ʧ����û�м�ʱֹͣͨ�������õ�SO2����___(ѡ���ƫ��������ƫ����������Ӱ����)
�������Ը��������Һ���ΪV1mL��Ũ��Ϊcmol/L�������������ӵ�ˮ�����ΪV2mL(������ɱ�״���µ����)����c��V1��V2��ʾSO2������ٷֺ���Ϊ___��
���𰸡�
������� | �������������� | ���� |
A | �������ơ�Ũ���� | ����SO2 |
B | Ũ���� | ��ȥˮ���� |
C | ||
D | ����������Һ | ���չ����Ķ�������ֹ������Ⱦ |
5SO2+2MnO4-+2H2O=5SO42-+2Mn2++4H+ ƫ�� ![]() ��100%
��100%
��������
��1���Ʊ������һ��װ�ã�����װ�á�����װ�á��ռ�װ�ã��ݴ˽��
��2��������Ʋ�����Լ�ѡ��������������Ũ�����Ʊ�����������Ķ���������Ҫ��װ��A�м����������ƣ���Һ©���м���Ũ���ᣬ���������к���ˮ������ͨ��Ũ�����ȥˮ�������ڼ���ƿ���ռ�������������������������������Һ���ն��������ֹ��Ⱦ������
��3���ٸ���������������Ի����¾���ǿ�������ԣ��ܹ�������������������������ӣ�
�ڷ�Ӧ������Һ�Ϻ�ɫ��ʧʱ��SO2�뷴Ӧ���������ĸ����������ǡ�÷�Ӧ����û��ʱֹͣͨ������δ��Ӧ��SO2����Ҳ��ˮ�������ܣ�
�۸��ݷ�Ӧ��5SO2+2MnO4-+2H2O=5SO42-+2Mn2++4H+��n��SO2��=2.5V1c��10-3mol������V=Vmn�����������������Ȼ������������İٷֺ�����
![]() �Ʊ������һ��װ�ã�����װ�á�����װ�á��ռ�װ�ã����ݷ�Ӧ����ʽ
�Ʊ������һ��װ�ã�����װ�á�����װ�á��ռ�װ�ã����ݷ�Ӧ����ʽ![]() ���Ʊ�����Ķ���������Ҫ��A�м����������ƹ��壬��Һ©���м���Ũ���ᣬ���ȷ�Ӧ���ɶ��������������к���ˮ��������Ҫͨ��Ũ�����ȥˮ�������ڼ���ƿ���������ſ������ռ��ռ���������Ķ����������������������Һ���չ����Ķ��������ֹ��Ⱦ�գ�װ����ͼ
���Ʊ�����Ķ���������Ҫ��A�м����������ƹ��壬��Һ©���м���Ũ���ᣬ���ȷ�Ӧ���ɶ��������������к���ˮ��������Ҫͨ��Ũ�����ȥˮ�������ڼ���ƿ���������ſ������ռ��ռ���������Ķ����������������������Һ���չ����Ķ��������ֹ��Ⱦ�գ�װ����ͼ ��
��
![]() ������Ʋ�����Լ�ѡ��������������Ũ�����Ʊ�����������Ķ���������Ҫ��װ��A�м��������ƣ���Һ©���м���Ũ���ᣬ���������к���ˮ������ͨ��Ũ�����ȥˮ�������ڼ���ƿ���ռ�������������������������������Һ���ն��������ֹ��Ⱦ��������д����Ϊ��
������Ʋ�����Լ�ѡ��������������Ũ�����Ʊ�����������Ķ���������Ҫ��װ��A�м��������ƣ���Һ©���м���Ũ���ᣬ���������к���ˮ������ͨ��Ũ�����ȥˮ�������ڼ���ƿ���ռ�������������������������������Һ���ն��������ֹ��Ⱦ��������д����Ϊ��
������� | �������������� | ���� |
A | �������ơ�Ũ���� | ���� |
B | Ũ���� | ��ȥˮ���� |
C | ||
D | ����������Һ | ���չ����Ķ�������ֹ������Ⱦ |
![]() ����������������Ի����¾���ǿ�������ԣ��ܹ�������������������������ӣ���Ӧ�����ӷ���ʽ��
����������������Ի����¾���ǿ�������ԣ��ܹ�������������������������ӣ���Ӧ�����ӷ���ʽ��![]() ��
��
![]() ��Ӧ������Һ�Ϻ�ɫ��ʧ����û�м�ʱֹͣͨ�����ͻ�ʹ��
��Ӧ������Һ�Ϻ�ɫ��ʧ����û�м�ʱֹͣͨ�����ͻ�ʹ��![]() �������
�������![]() �������������ĺ���ƫ�ͣ�
�������������ĺ���ƫ�ͣ�
![]() ���ݷ�Ӧ��
���ݷ�Ӧ��![]() ��
��![]() �����������
�����������![]() ����
����![]() ������
������![]() ������ٷֺ���Ϊ
������ٷֺ���Ϊ![]() ��
��

����Ŀ��NH4Al(SO4)2��ʳƷ�ӹ�����Ϊ��ݵ�ʳƷ���Ӽ������ڱ���ʳƷ����ҵ�ϳ�����������Ҫ�ɷ�ΪAl2O3��������SiO2��Fe2O3���ʣ��������������NH4Al(SO4)212H2O���乤������ͼ���£�
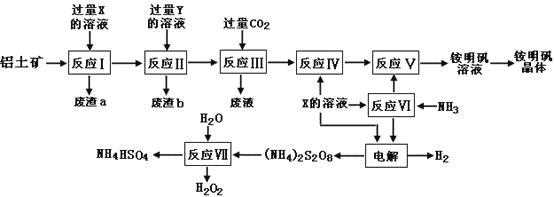
��1������a��b �ijɷֱַ��ǣ�_________��_____________����д���ƣ�
��2������ͼ��X�Ļ�ѧʽΪ��_______________��
��3����Ӧ������ӷ���ʽΪ��_________________________________________�����������Һ�л�����������IJ�������Ϊ����������ƣ�_________����ȴ�ᾧ������ϴ�ӡ�
��4���������[(NH4)2S2O8]�ڹ�ҵ�������й㷺����;��������Ϊ��������Ư�����㷺���������ع�ҵ���������ۺϵ�����������ά��ҵ���ѽ������������Ĺ����������ö��Ե缫���X�뷴Ӧ���������ʵĻ����Һ���Եõ�������李�
д��������Ӧʽ��______________________________________________��
��5����Ӧ���Ļ�ѧ����ʽΪ��________________________________________��
NH4HSO4��Һ������Ũ���ɴ�С˳��Ϊ��_____________________________��
��6�������������Һ����μ�������������Һ�������ܷ����ķ�Ӧ��______����ѡ����ĸ��
A��4NH4Al(SO4)2+3Ba(OH)2��2(NH4)2SO4+3BaSO4��+ Al2(SO4)3+2Al(OH)3�� |
B��2NH4Al(SO4)2+4Ba(OH)2��(NH4)2SO4+3BaSO4��+Ba(AlO2)2 |
C��2NH4Al(SO4)2+3Ba(OH)2��(NH4)2SO4+3BaSO4��+2Al(OH)3�� |
D��NH4Al(SO4)2+2Ba(OH)2��NH3��H2O+2BaSO4��+ Al(OH)3�� |