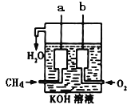
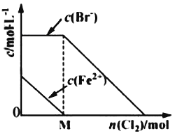
��Ŀ����
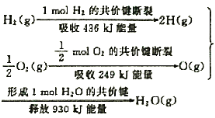
����Ŀ����֪��ѧ��Ӧ�е������仯,���ɻ�ѧ��Ӧ�оɻ�ѧ������ʱ���յ��������»�ѧ���γ�ʱ�ų���������ͬ���¡���ͼΪN2(g)��O2(g)����NO(g)�����е������仯��
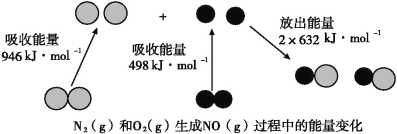
��1������ͨ���Ѳ�1 molij��ѧ�������յ��������ɸû�ѧ���ļ��ܡ����ܵĴ�С���Ժ�����ѧ����ǿ������N��N���ļ���Ϊ_________________kJ��mol-1��������ͼд��N2(g)��O2(g)����NO(g)���Ȼ�ѧ����ʽ��________________________________��
��2��NO(g)�ֽ�����N2(g)��O2(g)���Ȼ�ѧ����ʽΪ��_____________________������16 g O2(g)����ʱ,�÷�Ӧ�зų�������Ϊ��_________________kJ��
���𰸡�946N2(g)+O2(g)2NO(g)����H=180 kJ��mol-12NO(g)N2(g)+O2(g)����H=��180 kJ��mol-190
��������
��1������ʾ��ͼ��֪��N��N���ļ���Ϊ946kJ/mol����Ӧ�Ⱦ��Ƕϼ����յ��������γɻ�ѧ�����ų��������IJ�ֵ����˸��ݼ��ܿ�֪���÷�Ӧ�ķ�Ӧ����H��946kJ/mol��498kJ/mol��2��632kJ/mol����180kJ/mol�����Ը÷�Ӧ���Ȼ�ѧ����ʽ��N2(g)+O2(g)��2NO(g)����H=��180 kJ��mol-1���ʴ�Ϊ��946��N2(g)+O2(g)2NO(g)����H=+180 kJ��mol-1��
��2������Ӧ�����ȷ�Ӧ�����淴Ӧ�Ƿ��ȷ�Ӧ������NO(g)�ֽ�����N2(g)��O2(g)���Ȼ�ѧ����ʽΪ2NO(g)��N2(g)+O2(g)����H=��180 kJ��mol-1��16g������0.5mol�����Էų���������180 kJ��mol-1��0.5mol��90kJ���ʴ�Ϊ��2NO(g)N2(g)+O2(g)����H=��180 kJ��mol-1�� 90��

 ��ǰ����ϵ�д�
��ǰ����ϵ�д�