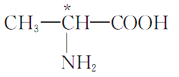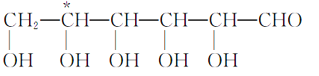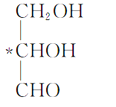��Ŀ����
����Ŀ��ʵ�����Ա���ȩΪԭ���Ʊ����屽��ȩ�ķ�Ӧ���£�
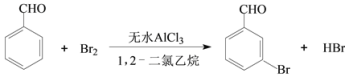
��֪����1�����屽��ȩ�¶ȹ���ʱ�ױ�������
��2���塢����ȩ��1,2-�������顢���屽��ȩ�ķе���±���
���� | �� | ����ȩ | 1,2-�������� | ���屽��ȩ |
�е�/�� | 58.8 | 179 | 83.5 | 229 |
����1����һ����ȵ���ˮAlCl3��1,2-��������ͱ���ȩ��ֻ�Ϻ�װ��������ƿ������ͼ��ʾ���������μӾ�Ũ��������������Һ�壬���·�Ӧһ��ʱ�䣬��ȴ��

����2������Ӧ����ﻺ������һ������ϡ�����У����衢���á���Һ���л�����10% NaHCO3��Һϴ�ӡ�
����3����ϴ�ӵ��л������������ˮMgSO4���壬����һ��ʱ�����˳�
MgSO4nH2O���塣
����4����ѹ�����л��㣬�ռ���Ӧ��֡�
��1��ʵ��װ���������ܵ���Ҫ������_____����ƿ��ӦΪ_____���ѧʽ����Һ��
��2������1��Ӧ�����У�Ϊ���ԭ�������ʣ����˵��¶ȷ�ΧΪ������ţ�_______��
A����229�� B��58.8��~179�� C����58.8��
��3������2����10% NaHCO3��Һϴ�ӣ���Ϊ�˳�ȥ�����л����___(�ѧʽ)��
��4������3�м�����ˮMgSO4�����������_____��
��5������4�в��ü�ѹ������Ϊ�˷�ֹ______��
��6����ʵ���м�����5.3 g����ȩ���õ�3.7 g���屽��ȩ������屽��ȩ����Ϊ______��
���𰸡��������� NaOH C Br2 ��ȥ�л����ˮ ���屽��ȩ���¶ȹ��߱����� 40%
��������
��1�������ܵ������ǣ����������������ԭ�������ʣ���ƿ��ʢ�ŵ���NaOH�������廯�⣻
��2������1��Ӧ�����У�Ϊ���ԭ�������ʣ�Ӧ�����¶�С����ķе㣻
��3������2���л�����10% NaHCO3��Һϴ��,����Ϊ�˳�ȥ�壻
��4������3�м�����ˮMgSO4���壬��Ϊ�˳�ȥ�л����е�ˮ��
��5�����ü�ѹ�����Խ��������ķе㣬��˿����ڽϵ͵��¶��·������֣���ֹ���屽��ȩ���¶ȹ��߱�������
��6�����ݹ�ϵʱ���㣬�����ϲ����屽��ȩ�ĵ�����������������ʡ�
��1�������ܵ������ǣ����������������ԭ�ϵ������ʣ����������廯��Ϊ�ж����壬��ƿ��ʢ�ŵ���NaOH��Һ�����������廯�⣻
��������������� ��NaOH��
��2������1��Ӧ�����У�Ϊ���ԭ�������ʣ�Ӧ�����¶ȵ�����ͱ���ȩ�ķе㣬�����¶�Ӧ����58.8�棻
�������C��
��3������2���л����к���δ��ȫ��Ӧ���壬������NaHCO3��Һ��Ӧ���ɿ�����ˮ���Σ������л�����10% NaHCO3��Һϴ��,����Ϊ�˳�ȥ�壻
���������
��4����ϴ�ӵ��л������������ˮMgSO4���壬����һ��ʱ�����˳�
MgSO4nH2O���壬���Բ���3�м�����ˮMgSO4���壬��Ϊ�˳�ȥ�л����е�ˮ��
�����Ϊ����ȥ�л����ˮ��
��5�����ü�ѹ�����Խ��������ķе㣬��˿����ڽϵ͵��¶��·������֣���ֹ���屽��ȩ���¶ȹ��߱�������
�����Ϊ�����屽��ȩ���¶ȹ��߱�������
��6������ ����5.3 g����ȩ��ȫ��Ӧ����x g���屽��ȩ,���У�106:185=5.3��x�����x=9.25g,���Լ��屽��ȩ����
����5.3 g����ȩ��ȫ��Ӧ����x g���屽��ȩ,���У�106:185=5.3��x�����x=9.25g,���Լ��屽��ȩ����![]() 100
100![]() =40
=40![]() ��
��
�������40![]() ��
��

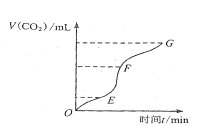
����Ŀ��ijͬѧΪ��̽��п�����ᷴӦ�����е����ʱ仯������100mLϡ�����м���������п�ۣ�����ˮ�������ռ���Ӧ�ų����������������������Ϊ��״���µ��������ʵ���¼���£��ۼ�ֵ����
ʱ��/min | 1 | 2 | 3 | 4 | 5 |
�������/mL | 50 | 120 | 232 | 290 | 310 |
��1����Ӧ��������ʱ�����__������0��1min����1��2min����2��3min������4��5min������ԭ����__��
��2����Ӧ������С��ʱ�����__������0��1min����1��2min����2��3min������4��5min������ԭ����__��
��3��2��3minʱ����ڣ��������Ũ�ȱ仯��ʾ�÷�Ӧ������Ϊ__��
��4�������Ӧ̫���ң�Ϊ�˼�����Ӧ���ʶ��ֲ����ٲ���������������ͬѧ�������зֱ��������������Һ�壬����Ϊ���е���__������ţ���
A.����ˮ B.NaCl��Һ C.Na2CO3��Һ D.CuSO4��Һ
����Ŀ��Ǧ�����ؼ۸������ԭ�����ã����÷�Χ�㡣��ŵ�ʱ�ķ�Ӧԭ��Ϊ��
Pb��PbO2��2H2SO4===2PbSO4��2H2O��
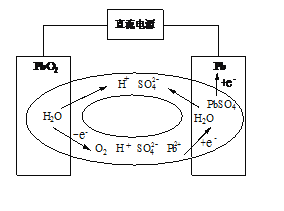
(1) д��Ǧ�����طŵ�ʱ�����ĵ缫��Ӧʽ��________���ŵ�ʱ��H+����______�������ƶ����������������
(2) ��ͳ��Ǧ�������ڳ��ĩ�ڣ��缫�ϻ����O2��Ϊ�˱��������ݳ��γ�������ʴ�豸����ѧ�ҷ������ܷ�Ǧ���أ�VRLA���������������շ��ﵽ�ܷ�Ч������ԭ����ͼ��ʾ����O2�����������յĵ缫��ӦʽΪ________��
(3) Ǧ�ĵ�⾫���ǹ�ҵ��ʵ�ַ�Ǧ�����Լ���Ǧ�ᴿ����Ҫ�ֶΡ�Ǧ�ĵ�⾫������PbSiF6��H2SiF6����ǿ�������ɵ�ˮ��Һ�н��С��ӻ�ԭ¯�в�����ij��Ǧ�ɷ����±���ʾ��
�ɷ� | Pb | Cu | Ag | Fe | Zn | Sn | ���� |
�� | 97.50 | 1.22 | 0.12 | 0.15 | 0.09 | 0.64 | 0.28 |
�ٵ�⾫��ʱ���������Ҫ�ɷ���________��Ԫ�ط��ţ���
�ڵ��������õ���Ǧ���Ժ����������ʣ����������п�����________����һ�֣���
�۵������У���Ǧ���������SiF6���壬д���õ缫��Ӧʽ________��