��Ŀ����
2���о����ʵĺϳɡ���ˮ�Ĵ���������ʵ����Ҫ���壮��1����ҵ�����У���NH3Ϊԭ�������ᣬд����ҵ�����������һ���Ļ�ѧ����ʽ3NO2+H2O=2HNO3+NO��
��2���ס��ҡ����������ʳ������������õ�ԭ�ϲ�ͬ��������������ͬ��
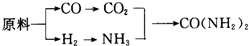
��֪��CO+H2O$\frac{\underline{\;����\;}}{\;}$CO2+H2��
����ҵ�йع涨������ԭ�����Ƶõ�ԭ����H2��CO2�����ʵ���֮�ȣ�����ӽ��ϳ����ص�ԭ����NH3�������H2�����ʵ�������CO2�����ʵ���֮�ȣ����ԭ�ϵ���������ߣ����мס��ҡ������ҹ�������ԭ�����£�
�׳��Խ�̿��ˮΪԭ�ϣ�
�ҳ�����Ȼ����ˮΪԭ�ϣ�
������ʯ���ͣ���Ҫ�ɷ�ΪC5H12����ˮΪԭ�ϣ�
�����������Ϣ�жϼס��ҡ������������ĸ�������ԭ�ϵ���������ߣ�������ס������ҡ���������
��3�������������в�����SO2ͨ���������̣�����ת��Ϊ��Ӧ�ü�ֵ������Ƶȣ�
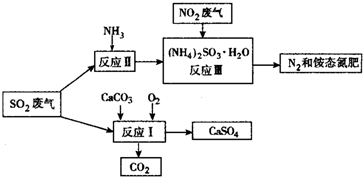
��д����Ӧ��Ļ�ѧ����ʽ��2CaCO3+O2+2SO2=2CaSO4+2CO2��
�������У���Ӧ�����Һ�м���ǿ��ԭ�ԵĶԱ����ӵ����ʣ�Ŀ���Ƿ�ֹ������隣�������
�ۼ��龭����Ӧ��õ����̬������SO42-�����Լ���������Ȼ�����
��4��Ϊ���ٻ�����Ⱦ����ҵ��Ҳ����Na2CO3��Һ������ȡHNO3������β���е�NO2������NaNO3��NaNO2��һ�����壬д����Ӧ�Ļ�ѧ����ʽ2NO2+Na2CO3=NaNO3+NaNO2+CO2��
���� ��1����NH3Ϊԭ�������ᣬ�Ȱ�������������NO����NO�������ɶ���������������������ˮ��Ӧ�������
��2����ԭ�����Ƶõ�H2��CO2�����ʵ���֮������ӽ��ϳ����ص�ԭ����NH3�������H2�����ʵ�������CO2�����ʵ���֮�ȣ�ԭ�ϵ���������ߣ�����ԭ����������H2��CO2�����ʵ���֮���жϣ�
��3���ٸ��ݷ�Ӧ��������д����Ӧ��Ļ�ѧ����ʽ��
�ڼ���ǿ��ԭ�ԵĶԱ����ӵ����ʣ�Ŀ���Ƿ�ֹ������泥�NH4��2SO3��������
�ۼ���SO42-�����Լ���������Ȼ�����
��4����Na2CO3��Һ������ȡHNO3������β���е�NO2������NaNO3��NaNO2��һ�����壬����ӦΪ������̼��
��� �⣺��1����NH3Ϊԭ�������ᣬ�Ȱ�������������NO����NO�������ɶ���������������������ˮ��Ӧ�������ᣬ��ҵ�����������һ���Ļ�ѧ����ʽΪ3NO2+H2O=2HNO3+NO���ʴ�Ϊ��3NO2+H2O=2HNO3+NO��
��2��NH3�������H2�����ʵ�������CO2�����ʵ���֮��Ϊ2��1�ϳ����أ���H2��CO2�����ʵ���֮��Ϊ$\frac{2��3}{2}$��1=3��1��
�׳��Խ�̿��ˮΪԭ�ϣ�C+2H2O$\frac{\underline{\;����\;}}{\;}$CO2+2H2�����ɵ�H2��CO2�����ʵ���֮��Ϊ2��1��
�ҳ�����Ȼ����ˮΪԭ�ϣ�CH4+2H2O$\frac{\underline{\;��ȼ\;}}{\;}$CO2+4H2�����ɵ�H2��CO2�����ʵ���֮��Ϊ4��1��
������ʯ���ͺ�ˮΪԭ�ϣ�C5H12+10H2O$\frac{\underline{\;����\;}}{\;}$5CO2+16H2�����ɵ�H2��CO2�����ʵ���֮��Ϊ16��5������ӽ�3��1��
�ʴ�Ϊ������
��3���ٸ��ݷ�Ӧ����������֪��Ӧ��Ļ�ѧ����ʽΪ��2CaCO3+O2+2SO2=2CaSO4+2CO2��
�ʴ�Ϊ��2CaCO3+O2+2SO2=2CaSO4+2CO2��
��������������ױ���������Ӧ�����Һ�м���ǿ��ԭ�ԵĶԱ����ӵ����ʣ�Ŀ���Ƿ�ֹ��NH4��2SO3��������
�ʴ�Ϊ����ֹ������隣�������
�ۼ���SO42-�����Լ���������Ȼ�����HCl �� BaCl2�����ʴ�Ϊ��������Ȼ�����
��4����Na2CO3��Һ������ȡHNO3������β���е�NO2������NaNO3��NaNO2��һ�����壬����ӦΪ������̼���÷�ӦΪ2NO2+Na2CO3=NaNO3+NaNO2+CO2��
�ʴ�Ϊ��2NO2+Na2CO3=NaNO3+NaNO2+CO2��
���� ���⿼����������ᴿ�ۺ�Ӧ�ã�Ϊ��Ƶ���㣬����ϰ������Ϣ�������������̵�Ϊ���Ĺؼ������ط�����Ӧ�á�ʵ���������ۺϿ��飬��Ŀ�ѶȲ���

| A�� | ����ԭ�ӵĵ��Ӳ���ȫ������s���� | |
| B�� | 3p�ܼ���ֻ��һ���չ����ԭ�Ӻ�3p�ܼ�����һ��δ�ɶԵ��ӵ�ԭ�� | |
| C�� | ���������Ų�ʽΪ2s22p6��ԭ�Ӻ����������Ų�ʽΪ2s22p6������ | |
| D�� | ԭ�Ӻ����M���ϵ�s�ܼ���p�ܼ��������˵��ӣ���d�������δ���е��ӵ�����ԭ�� |
| A�� | 0.15 mol/L | B�� | 0.20 mol/L | C�� | 0.25 mol/L | D�� | 0.40 mol/L |
| A�� | NaHCO3�ĵ��룺NaHCO3?Na++H++CO32- | |
| B�� | HS-��ˮ�⣺HS-+H2O?H3O++S2- | |
| C�� | ����ı�ȼ����Ϊ-890.3kJ•mol-1�������ȼ�յ��Ȼ�ѧ����ʽ�ɱ�ʾΪ��CH4��g��+2O2��g���TCO2��g��+2H2O ��g����H=-890.3kJ•mol-1 | |
| D�� | ˫��ˮ�м���ϡ�����KI��Һ��H2O2+2I-+2H+�TI2+2H2O |
| A�� | ��������������Һ��¶�ڿ����У�4Fe2++O2+4H+�T4Fe3++2H2O | |
| B�� | ����ʯ��ˮ��̼����Ʒ�Ӧ��Ca2++2HCO${\;}_{3}^{-}$+2OH-�TCaCO3��+CO${\;}_{3}^{2-}$+2H2O | |
| C�� | ��ƫ��������Һ��ͨ�����CO2��AlO2-+CO2+2H2O�TAl��OH��3��+HCO${\;}_{3}^{-}$ | |
| D�� | ��������ϡ���ᷴӦ��3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O |
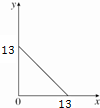 ij�¶ȣ�T�棩�µ���Һ�У�c��H+��=10-x mol•L-1��c��OH-��=10-y mol•L-1��x��y�Ĺ�ϵ��ͼ��ʾ����ش��������⣺
ij�¶ȣ�T�棩�µ���Һ�У�c��H+��=10-x mol•L-1��c��OH-��=10-y mol•L-1��x��y�Ĺ�ϵ��ͼ��ʾ����ش��������⣺��1�����¶�T��25�棨���������������=������
��2���ڴ��¶��£���Ba��OH��2��Һ����μ���pH=a�����ᣬ��û����Һ�IJ���pH�����ʾ��
| ʵ�� ��� | Ba��OH��2��Һ �����/mL | ��������/mL | ��Һ��pH |
| �� | 22.00 | 0.00 | 9 |
| �� | 22.00 | 18.00 | b |
| �� | 22.00 | 22.00 | 6 |
��3���ڴ��¶��£���10mL 0.1mol•L-1��Ba��OH��2��Һ�ֱ���5mL 0.1mol•L-1��NaHSO4��Һ��ϣ�������Һ��pHΪ12���������Ũ�ȵ������Ba��OH��2��Һ��NH4Cl��Һ��ϣ�������Һ�и�����Ũ���ɴ�С������˳����c��OH-����c��Ba2+��=c��Cl-����c��NH4+����c��H+����
 ��
��