��Ŀ����
��11�֣��Ӻ�ˮ�п��Ի�õ�ˮ��ʳ�Σ�������ȡþ��������ʡ�
��һ����ˮ�����ķ�����Ҫ�� ��ֻ��һ�֣���
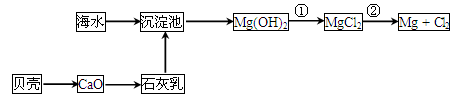
�������Ӻ�ˮ����ȡ���þ����������ͼ��ʾ��
��1����ȡBr2ʱ����һ��ͨ��Cl2������Ӧ�����ӷ���ʽ�� ��
����SO2���� �����������ԭ�����ԣ�
�ڶ���ͨ��Cl2��Ҫ��ȡBr2��Ҫ���еIJ���Ϊ ��
��2��Ϊ��ʵ�ֶ�þ���ӵĸ��������м���������Լ��� ���ѧʽ����
��ӽ�Լ��Դ����߽���þ�Ĵ��ȷ��������������˵�ұ���ķ�����
������ĸ����
A��Mg(OH)2 MgO MgO  Mg Mg |
B��Mg(OH)2  MgO MgO  Mg Mg |
C��Mg(OH)2 ��ˮMgCl2 ��ˮMgCl2 Mg Mg |
D��Mg(OH)2 MgCl2����Һ�� MgCl2����Һ�� Mg Mg |
��һ��������������1��Cl2 + 2Br��= 2Cl��+ Br2 ����ԭ��1�֣�����ȡ����Һ��
��2��Ca(OH)2����NaOH�����������𰸾����֣���C ��
���������������һ����ˮ�����ķ�����Ҫ��������������1����ȡBr2ʱ����һ��ͨ��Cl2������Ӧ�����ӷ���ʽ��Cl2 + 2Br��= 2Cl��+ Br2������SO2�е�SԪ�صĻ��ϼ����ߣ�ʧȥ���ӱ��ֳ���ԭ�ԣ��ڶ���ͨ��Cl2��Ҫ��ȡBr2�������嵥�����л��ܼ��������ܽ⣬���Եõ���Ũ�ȵ�Br2����Ҫ���еIJ���Ϊ��ȡ����Һ����2��Ϊ��ʵ�ֶ�þ���ӵĸ��������м���������Լ���Ca(OH)2����NaOH�ȣ��������ʣ��ӽ�Լ��Դ����߽���þ�Ĵ��ȷ��������������˵�ұ���ķ����ǵ�����ڵ�MgCl2�ķ��������ѡ����C��
���㣺���麣ˮ��Դ���ۺ����õ�֪ʶ��

��ˮ��һ����Ļ�ѧ��Դ�⣬�����йغ�ˮ�ۺ����õ�˵����ȷ���ǣ� ��
| A����ˮ�к��м�Ԫ�أ�ֻ�辭�������仯���ܵõ��ص��� |
| B����ˮ�����ƺ��εĹ�����ֻ�����˻�ѧ�仯 |
| C���Ӻ�ˮ�п��Եõ�NaCl���������NaCl���Ʊ�Na |
| D�����ó�ϫ�����ǽ���ѧ��ת��Ϊ���� |
�����й���Դ�Ŀ�������˵����ȷ���� �� ��
| A���Ӻ�������ȡ�ⵥ�ʵĹ���һ���漰������ԭ��Ӧ |
| B���Ӻ�ˮ�п��Եõ�NaCl�����NaCl��Һ���Ʊ�Na��Cl2 |
| C����Ȼ�����Ҵ���ˮú���ֱ����ڻ�ʯ��Դ������������Դ�Ͷ�����Դ |
| D��Cu��Al��Hg���Էֱ����Ȼ�ԭCuO�����AlCl3���ȷֽ�HgOұ���õ� |
��������ۺ����ü������ڽ�Լ��Դ���������ڱ���������ʵ�������÷Ͼɻ�ͭ(Cu��Zn�Ͻ𣬺���������Fe)�Ʊ���������(CuSO4��5H2O)��������ZnO���Ʊ�����ͼ���£� 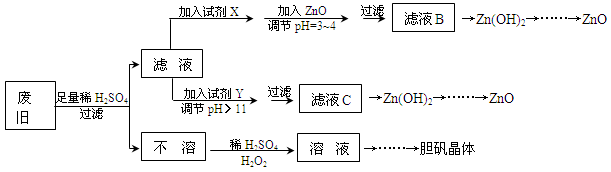
��֪��Zn���������������Al����������������ƣ�pH��11ʱZn(OH)2������NaOH��Һ����[Zn(OH)4]2�����±��г��˼������������������������pH(��ʼ������pH����������Ũ��Ϊ1.0mol��L��1����)��
| | Fe3�� | Fe2�� | Zn2�� |
| ��ʼ������pH | 1.1 | 5.8 | 5.9 |
| ������ȫ��pH | 3.0 | 8.8 | 8.9 |
��ش��������⣺
��1���Լ�X������__________����������____________________��
��2������ZnO����pH=3��4��Ŀ����____________________��
��3���ɲ�����������ҺD�Ļ�ѧ����ʽΪ______________________________��
��4������ҺD�Ƶ��������������Ҫ����������______________________________��
��5�������Լ�����ΪY�Լ�����______��
A��ZnO B��NaOH C��Na2CO3 D��ZnSO4
������ҺC����μ�������ֱ���������������������______________________________��
��6���ⶨ��������Ĵ���(��������I��������Ӧ������������)��ȷ��ȡ0.5000g��������������ƿ�У�������ˮ�ܽ⣬�ټ������KI����0.1000mol��L��1Na2S2O3����Һ�ζ����յ㣬����Na2S2O3����Һ19.40mL����֪�������ζ������е����ӷ���ʽ���£�
2Cu2����4I��
 2CuI(��ɫ)����I2��I2��2S2O32��
2CuI(��ɫ)����I2��I2��2S2O32�� 2I����S4O62��
2I����S4O62���ٵ�������Ĵ���Ϊ_______________��
���ڵζ������о���ҡ��(��Һ���⽦)��ƿ��������õĴ��Ƚ���__________(�ƫ�ߡ�����ƫ�͡����䡱)��
(14��)ﯲ�ҵ�Ǽ��з�չDZ����ǰ�������˲�ҵ���(Zr)Ԫ���Ǻ˷�Ӧ��ȼ�ϰ��İ������ϣ��������(ZrO2)�����������������մɡ��ҹ��зḻ���Ӣʯ(ZrSiO4)����Al2O3��SiO2��Fe2O3�����ʣ����������֮һ���£�
�Իش��������⣺
(1)д�����������и��������ķ�Ӧ����ʽ(̼ת����CO)��_______________________________________________��
(2)д��ZrOCl2��8H2O��900 ������ZrO2�ķ�Ӧ����ʽ�� ______________ ______________________________________________��
(3)���ڶ�����������մɺ�ﯺϽ��˵������ȷ����________(��ѡ)��
| A��������������մ����������ǽ������� |
| B��1 nm��10��10m |
| C��ﯺϽ��Ӳ�ȱȴ��Ҫ�� |
| D���ձ������˵�վ�ı�ը��������ﯺϽ��ڸ�������ˮ������Ӧ���������� |
(4)һ������ȼ�ϵ�أ�һ��ͨ���������һ��ͨ�붡�飻������Dz���������(Y2O3)�Ķ������(ZrO2)���壬������״̬���ܴ���O2���������ڵ�����У�O2����________(���������)���ƶ�����������缫��ӦΪ__________________�������缫��ӦΪ________________________________��
���ݺ�ˮ�ۺ����õĹ�������ͼ���ж�����˵����ȷ����
�� ��
��֪��MgCl2��6H2O��������Mg��OH��Cl��HCl����ȡ�
| A�����̢ٵ��ᴿ���������̣����̢�ͨ��������ԭ��Ӧ�ɲ���2�ֵ��� |
| B���ڹ��̢��н�MgCl2��6H2O���ռ����Ƶ���ˮMgCl2 |
| C�����̢ݷ�Ӧ����Һ��ǿ���ԣ���������������豸�ĸ�ʴ���� |
| D���ڹ��̢ܡ���Ӧ��ÿ����0.2 mol Br-������2. 24 L Cl2 |