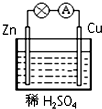
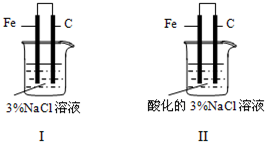
��Ŀ����
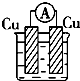
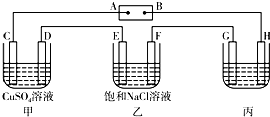
��ͼ��ʾ��װ�ã�C��D��E��F��Ϊ���Ե缫������Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ���Իش��������⣺��1���缫A��______���������������
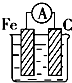
��2����װ����C���ĵ缫��ӦʽΪ______��
��3�����ö��Ե缫��ⱥ��NaCl��Һһ��ʱ�䣬����������56 mL����״���£����壬������Һ���Ϊ500 mLʱ��������Һ��25��ʱ��pH=______��
��4�����ñ�װ�ø�ͭ������GӦ����______���ͭ���������������Һ����Ҫ�ɷ��ǣ��ѧʽ��______��
��2����װ����C���ĵ缫��ӦʽΪ______��
��3�����ö��Ե缫��ⱥ��NaCl��Һһ��ʱ�䣬����������56 mL����״���£����壬������Һ���Ϊ500 mLʱ��������Һ��25��ʱ��pH=______��
��4�����ñ�װ�ø�ͭ������GӦ����______���ͭ���������������Һ����Ҫ�ɷ��ǣ��ѧʽ��______��

��1����װ���У�����Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ��˵��F�缫�������ӷŵ磬ͬʱ�缫�������������������ɣ���FΪ������B��ԭ��ظ�������A��ԭ����������ʴ�Ϊ��������
��2������C�缫�����������ӷŵ磬�缫��ӦʽΪ4OH--4e-=2H2O+O2�����ʴ�Ϊ��4OH--4e-=2H2O+O2����
��3���ö��Ե缫��ⱥ��NaCl��Һʱ�������������ӷŵ�������������ط�ӦʽΪ��2NaCl+2H2O
2NaOH+Cl2��+H2��������������NaOH֮��Ĺ�ϵʽ��n��NaOH��=2n��Cl2��=
��2=0.005mol��c��OH-��=c��NaOH��=
=0.01MOL/l��
c��H+��=
mol/L=10-12 mol/L������Һ��pH=12���ʴ�Ϊ��12��
��4�����ʱ���Ʋ����������Ƽ����������������ҺΪ�����Ե����Σ���װ�ø�ͭ������GӦ�����������Һ����Ҫ�ɷ���AgNO3���ʴ�Ϊ������AgNO3��
��2������C�缫�����������ӷŵ磬�缫��ӦʽΪ4OH--4e-=2H2O+O2�����ʴ�Ϊ��4OH--4e-=2H2O+O2����
��3���ö��Ե缫��ⱥ��NaCl��Һʱ�������������ӷŵ�������������ط�ӦʽΪ��2NaCl+2H2O
| ||
| 0.056L |
| 22.4L/mol |
| 0.005mol |
| 0.5L |
c��H+��=
| 10-14 |
| 0.01 |
��4�����ʱ���Ʋ����������Ƽ����������������ҺΪ�����Ե����Σ���װ�ø�ͭ������GӦ�����������Һ����Ҫ�ɷ���AgNO3���ʴ�Ϊ������AgNO3��

��ϰ��ϵ�д�
�����Ŀ