��Ŀ����
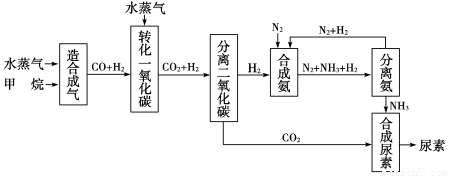
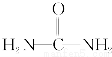
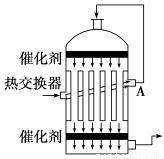
��ͼ��ʾ���Ǻ����Ƽ��������ʾ��ͼ��
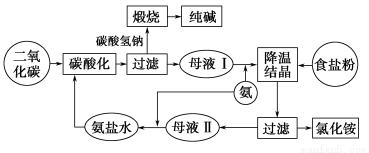
��ش�
(1)ĸҺ�������ܽ����Ҫ������________(д���ʵĻ�ѧʽ)��ĸҺ�������е�Ũ�ȵ�Na2CO3����Ҫԭ����__________________________
(2)��ĸҺ����ͨ������ʱҪ��ͨ������ͨ������̼���壬��Ҫԭ����_______________________________________
(3)ĸҺ����ĸҺ���ж�ͨ��NH3����NH3����Ҫ��Դ��__________����ĸҺ����ͨ��NH3����ҪĿ����____________����ĸҺ����ͨ��NH3����ҪĿ����__________________��
(4)�������̵������������������������____________��ѭ����
(1)NaCl��NaHCO3��NH4Cl��HCO3�� �����CO32��
(2)NH3��NaCl��Һ���ܽ�Ⱥܴ�CO2�ڰ�������Һ���ܽ�ȴ���ͨNH3��ͨCO2���γɸ�Ũ�ȵ�HCO3��������ͨCO2��ͨNH3������CO2��NaCl��Һ���ܽ�Ⱥ�С������NH3��̼�ữ����Һ���ܽ�ȴ�Ҳ�����γɸ�Ũ�ȵ�HCO3��
(3)���ϳɰ�����������NH4����Ũ�ȣ��ٽ�NH4Cl�ᾧ������������OH����Ũ�ȣ��ٽ�CO2���ܽ⣬����HCO3����Ũ��
(4)������̼(��CO2)
�������������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���ˮ��þ���ܴ���ԼΪ2.1��1015t��Ŀǰ������60%��þ���Ժ�ˮ����ҵ��ģ��ˮ��þ��������ͼ��ʾ��
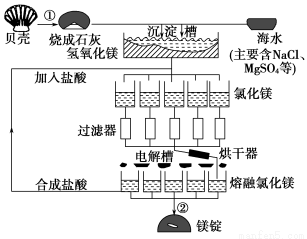
�Իش��������⣺
(1)��ˮ��þ�Ĺ�������������Ӧ�Ļ�ѧ����ʽ��
��_____________________________________��
��______________________________________��
(2)���Ȼ�þ��Һ��ȡ��ˮ�Ȼ�þ���壬�����������____________________
(3)Ϊ��ʹMgSO4��ȫת��ΪMg(OH)2����������ʯ��Ҫ������Ȼ������Mg(OH)2����������Ca(OH)2�ܽ�ȣ�Ӧ����________�����롣
(4)����þ�����Ĺ�ҵұ��������������֮�������в�֮ͬ���±�������þ���Ȼ�þ���۷е����ݣ�
���� | ����þ | �Ȼ�þ |
�۵�(��) | 2 852 | 714 |
�е�(��) | 3 600 | 1 412 |
��ҵ����þ���õ�������Ȼ�þ����ұ�������õ�����ڵ�Al2O3����ԭ����____________________________________