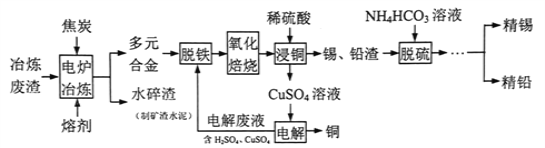
��Ŀ����
����Ŀ��NOx�ǿ�������Ҫ��Ⱦ��֮һ���ش��������⣺
(1)����β���е�NO�����ڷ�Ӧ��N2(g)�� O2(g)![]() 2NO(g)����ͼ��ʾ�ڲ�ͬ�¶�ʱ��һ����NO�ֽ������N2�����������ʱ��ı仯����N2(g)�� O2(g)
2NO(g)����ͼ��ʾ�ڲ�ͬ�¶�ʱ��һ����NO�ֽ������N2�����������ʱ��ı仯����N2(g)�� O2(g)![]() 2NO(g)Ϊ________(��������������������)��Ӧ��2000��ʱ�����ݻ�Ϊ2L���ܱ������г���10mol N2��5mol O2������������Ӧ��10min�ﵽƽ�⣬����2mol NO����10min�ڵ�ƽ����Ӧ������(O2)��____mol��L��1��min��1��
2NO(g)Ϊ________(��������������������)��Ӧ��2000��ʱ�����ݻ�Ϊ2L���ܱ������г���10mol N2��5mol O2������������Ӧ��10min�ﵽƽ�⣬����2mol NO����10min�ڵ�ƽ����Ӧ������(O2)��____mol��L��1��min��1��

(2)���������������ϼ�װ��ת��װ�ÿɼ���NOx���ŷš��о�������NOx���ѳ��ʳ��뻹ԭ������������⣬��ȡ���ڴ���������ȱλ���ܼ��̶ȡ���La0.8A0.2BCoO3��X(A��B��Ϊ����Ԫ��)Ϊ��������H2��ԭNO�Ļ������£�
��һ�Σ�B4��(���ȶ�)��H2���ͼ�̬�Ľ�������(��ԭǰ������н���ԭ�ӵĸ�������)
�ڶ��Σ�NO(g)������NO(a) ��H1��K1
2NO(a)��2N(a)+O2(g) ��H2��K2
2N(a)��N2(g)��2�� ��H3��K3
2NO(a)��N2(g)+2O(a) ��H4��K4
2O(a)��O2(g)��2�� ��H5��K5
ע������ʾ�����������ȱλ��g��ʾ��̬��a��ʾ����̬
��һ����������ԭB4���õ��ͼ�̬�Ľ�������Խ�࣬�ڶ��η�Ӧ������Խ�죬ԭ����_____���ڶ����и���Ӧ�ʱ��Ĺ�ϵ��2��H1����H2����H3��________�����¶��£�NO�ѳ���Ӧ2NO(g)![]() N2(g)��O2(g)��ƽ�ⳣ��K��_______(��K1��K2��K3�ı���ʽ��ʾ)��
N2(g)��O2(g)��ƽ�ⳣ��K��_______(��K1��K2��K3�ı���ʽ��ʾ)��
(3)��ҵ�����е�NO��NO2����NaOH��Һ���ա���20mL0.1mo1��L��1NaOH��Һ��ͨ�뺬NO��NO2�ķ�������ҺpH���������V�ı仯��ͼ1��ʾ(�������¶ȱ��ֲ���)��B���Ӧ����Һ��c(HNO2)��c(NO2��)����A���Ӧ��Һ��c(NO2-)/c(HNO2)��________��

(4)������ԭ��Ҳ��������NO��ԭ����ͼ2��ʾ��NO����ת��ΪH2O��________(�����ʽ)��������1 molNH3��0.5 molO2ʱ�������Ͽ�����____________L(��״��)NO��
���𰸡����� 0.05 ��ԭ������н���ԭ�ӵĸ������䣬��̬���ͣ���ȱλ���࣬��Ӧ���ʼӿ� 2��H1����H4����H5 K12��K2��K3 103.7 ![]() 11.2
11.2
��������
(1)����ͼ��T2�����ȵ���ƽ�⣬��Ӧ���ʴ��¶Ƚϸߣ�����¶ȶ�ƽ���Ӱ������жϣ�����N2(g)�� O2(g)![]() 2NO(g)���������Ӧ���ĵ��������ټ���10min�ڵ�������ƽ����Ӧ���ʣ�
2NO(g)���������Ӧ���ĵ��������ټ���10min�ڵ�������ƽ����Ӧ���ʣ�
(2)��ԭ������н���ԭ�ӵĸ������䣬��̬���ͣ���ȱλ���ࣻ���ݸ�˹���ɷ������
(3)NO��NO2����NaOH��Һ�����շ����ķ�ӦΪNO+NO2+2NaOH�T2NaNO2+H2O�������������Ƶ�ˮ��ƽ�ⳣ��Kh= ����¶Ȳ��䣬Kh������㣻
����¶Ȳ��䣬Kh������㣻
(4)��ͼ2֪����ԭ���е���NO����ת��ΪH2O�͵��������ݰ���ʧȥ�ĵ��ӵ����ʵ�������NO�������õ��ĵ��������ʵ������㡣
(1)����ͼ��T2�����ȵ���ƽ�⣬��Ӧ���ʴ��¶Ƚϸߣ����¶����ߣ����������������С��˵�������¶�ƽ��������Ӧ�����ƶ���������ӦΪ���ȷ�Ӧ������N2(g)�� O2(g)![]() 2NO(g)��10min�ﵽƽ�⣬����2mol NO����Ӧ������Ϊ1mol��10min�ڵ�ƽ����Ӧ������(O2)��
2NO(g)��10min�ﵽƽ�⣬����2mol NO����Ӧ������Ϊ1mol��10min�ڵ�ƽ����Ӧ������(O2)��![]() =0.05 mol��L��1��min��1���ʴ�Ϊ�����ȣ�0.05��
=0.05 mol��L��1��min��1���ʴ�Ϊ�����ȣ�0.05��
(2)��ԭ������н���ԭ�ӵĸ������䣬��̬���ͣ���ȱλ���࣬�ڶ��η�Ӧ���ʼӿ죻�ڶ��Σ���NO(g)������NO(a) ��H1��K1����2NO(a)��2N(a)+O2(g) ��H2��K2����2N(a)��N2(g)��2�� ��H3��K3����2NO(a)��N2(g)+2O(a) ��H4��K4����2O(a)��O2(g)��2�� ��H5��K5�����ݸ�˹���ɣ�������2+��+�۵õ�2NO(g)��O2(g)+ N2(g)��������2+��+�ݵõ�2NO(g)��N2(g)+ O2(g)����2��H1����H2����H3��2��H1����H4����H5�����2NO(g)![]() N2(g)��O2(g)��ƽ�ⳣ��K��K12��K2��K3���ʴ�Ϊ����ԭ������н���ԭ�ӵĸ������䣬��̬���ͣ���ȱλ���࣬��Ӧ���ʼӿ죻2��H1����H4����H5��K12��K2��K3��
N2(g)��O2(g)��ƽ�ⳣ��K��K12��K2��K3���ʴ�Ϊ����ԭ������н���ԭ�ӵĸ������䣬��̬���ͣ���ȱλ���࣬��Ӧ���ʼӿ죻2��H1����H4����H5��K12��K2��K3��
(3)20mL0.1mo1��L��1NaOH��Һ�к�����������0.002mol��NO��NO2����NaOH��Һ���գ������ķ�ӦΪNO+NO2+2NaOH�T2NaNO2+H2O���������Ƶ�ˮ��ƽ�ⳣ��Kh= ��B���Ӧ����Һ��c(HNO2)��c(NO2��)��Kh= c(OH-)=
��B���Ӧ����Һ��c(HNO2)��c(NO2��)��Kh= c(OH-)=![]() =10-10.7���¶Ȳ��䣬Kh���䣬A��Kh=
=10-10.7���¶Ȳ��䣬Kh���䣬A��Kh= ��
��![]() =
= =
=![]() =103.7���ʴ�Ϊ��103.7��
=103.7���ʴ�Ϊ��103.7��
(4)��ͼ2��֪��Ӧ��Ϊ������һ�������Ͱ�������������Ϊ������ˮ������NO����ת��ΪN2��H2O�������ĵ���ʽΪ![]() ��������һ�������Ͱ�����Ӧ���ɵ�����ˮ����Ӧ�а���ʧȥ�ĵ��ӵ����ʵ�������NO�������õ��ĵ��������ʵ�����1mol NH3ת��ΪN2ʧȥ3mol���ӣ�0.5mol O2�õ�2mol���ӣ���NOת��ΪN2�õ��ĵ���Ϊ1mol������NO�����ʵ���Ϊ0.5mol����״���µ����Ϊ11.2L���ʴ�Ϊ��
��������һ�������Ͱ�����Ӧ���ɵ�����ˮ����Ӧ�а���ʧȥ�ĵ��ӵ����ʵ�������NO�������õ��ĵ��������ʵ�����1mol NH3ת��ΪN2ʧȥ3mol���ӣ�0.5mol O2�õ�2mol���ӣ���NOת��ΪN2�õ��ĵ���Ϊ1mol������NO�����ʵ���Ϊ0.5mol����״���µ����Ϊ11.2L���ʴ�Ϊ��![]() ��11.2��
��11.2��

 ������ÿ�ʱ��ҵϵ�д�
������ÿ�ʱ��ҵϵ�д�����Ŀ������ʵ������淶���ܴﵽĿ�ĵ��ǣ� ��
Ŀ�� | ���� | |
A | ����480mL0.1mol/LCuSO4��Һ | ��ȡ8.0g��ˮ����ͭ�����ձ��У�����480mL������ˮ���ò��������衣 |
B | ��ȥ�Ȼ��ƹ�����������̼��� | ��ˮϡ���ᡢȻ��������ɡ� |
C | ��ȡ��ˮ�еĵ� | ����Ͳ��ȡһ�����ĵ�ˮ�������Һ©����ע�����Ȼ�̼�������ú��Һ�� |
D | ��ȥN2�����е�����O2 | ��ȼ��Ȼ�������ͨ�����ʯ��ˮ |
A.AB.BC.CD.D