��Ŀ����
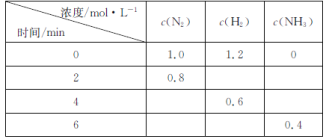
����Ŀ����һ����̶����ܱ������м��뷴Ӧ��N2��H2��������Ӧ��N2(g)+3H2(g)![]() 2NH3(g)����Ӧ�����еIJ������������ʾ��
2NH3(g)����Ӧ�����еIJ������������ʾ��
����˵����ȷ����
A.0��2min�ڣ�H2�ķ�Ӧ����Ϊ0.1 mol/(L��min)
B.2minʱ��NH3�����ʵ���Ũ��Ϊ0.2 mol/L
C.4minʱ����Ӧ�Ѵﵽƽ��״̬����ʱ�����淴Ӧ�����ʶ�Ϊ0
D.4��6min�ڣ�������������ӵ������ʵ�������
���𰸡�D
��������
A. ���ݱ������ݿ�֪�����ڷ�ӦN2(g)+3H2(g)![]() 2NH3(g)���ڿ�ʼʱc(N2)=1.0 mol/L��c(H2)=1.2 mol/L��c(NH3)=0 mol/L����2 minʱc(N2)=0.8 mol/L���ı���0.2 mol/L�������N2��H2�ķ�Ӧ��ϵ��֪��ͬʱ����H2 0.6 mol/L������0��2min�ڣ�H2�ķ�Ӧ����Ϊv (N2)=
2NH3(g)���ڿ�ʼʱc(N2)=1.0 mol/L��c(H2)=1.2 mol/L��c(NH3)=0 mol/L����2 minʱc(N2)=0.8 mol/L���ı���0.2 mol/L�������N2��H2�ķ�Ӧ��ϵ��֪��ͬʱ����H2 0.6 mol/L������0��2min�ڣ�H2�ķ�Ӧ����Ϊv (N2)=![]() =0.3 mol/(L��min)��A����
=0.3 mol/(L��min)��A����
B. ���ݱ������ݿ�֪2 minʱN2Ũ�ȱ仯0.2 mol/L�����ڷ�ӦʱN2��NH3Ũ�ȸı�ı�Ϊ1��2������NH3��Ũ�ȱ仯��0.4 mol/L����ʼʱNH3��Ũ��Ϊ0����2 minʱ��NH3�����ʵ���Ũ��Ϊ0.4 mol/L��B����
C. 4 minʱH2��Ũ�ȱ仯��(1.2-0.6)mol/L=0.6 mol/L����ʱ����NH3��Ũ�ȱ仯��0.4 mol/L����6 min��Ũ����ͬ��˵����Ӧ�ﵽƽ�⣬��ʱ�����淴Ӧ�����ʶ���ȣ�����Ϊ0��C����
D. ����ѡ��C������֪����4��6 min����Ӧ�ﵽƽ��״̬���κ����ʵ����ʵ������䣬������������ӵ������ʵ������䣬D��ȷ��
�ʺ���ѡ����D��

 ȫ�ſ��䵥Ԫ�����������ܸ�ϰϵ�д�
ȫ�ſ��䵥Ԫ�����������ܸ�ϰϵ�д� Ʒѧ˫�ž�ϵ�д�
Ʒѧ˫�ž�ϵ�д� Сѧ��ĩ���100��ϵ�д�
Сѧ��ĩ���100��ϵ�д�����Ŀ����̼����������ࡣ�ش��������⣺
(1)һ���¶��£����ܱ������н������ʵ�����CO(g)��H2O(g)��ϣ������ʵ��Ĵ������з�ӦCO(g)+H2O(g)![]() CO2(g)+H2(g)����֪���¶��£��÷�Ӧ��ƽ�ⳣ��K=16����ƽ��ʱ��ϵ��H2�����ʵ�������Ϊ_________%��
CO2(g)+H2(g)����֪���¶��£��÷�Ӧ��ƽ�ⳣ��K=16����ƽ��ʱ��ϵ��H2�����ʵ�������Ϊ_________%��
(2)�ڴ���Ru���£�CO2��H2��Ӧ������CH4����Ӧ����ʽΪCO2(g)+4H2(g)![]() CH4(g)+2H2O(g)����֪H2������������¶ȵ����߶����ӡ����¶ȴ�300������400�棬���´ﵽƽ�⣬��v��______(��������������С����������������ͬ)��v��_______��ƽ�ⳣ��K________��ת������________��������ͬ�¶�ʱ��������Ӧ�ڲ�ͬ��ʼŨ���·ֱ�ﵽƽ�⣬�����ʵ�ƽ��Ũ�����±���
CH4(g)+2H2O(g)����֪H2������������¶ȵ����߶����ӡ����¶ȴ�300������400�棬���´ﵽƽ�⣬��v��______(��������������С����������������ͬ)��v��_______��ƽ�ⳣ��K________��ת������________��������ͬ�¶�ʱ��������Ӧ�ڲ�ͬ��ʼŨ���·ֱ�ﵽƽ�⣬�����ʵ�ƽ��Ũ�����±���
c(CO2)/mol/L | c(H2)/mol/L | c(CH4)/mol/L | c(H2O)/mol/L | |
ƽ��I | a | b | c | d |
ƽ��II | m | n | x | y |
��a��b��c��d��m��n��x��y֮��Ĺ�ϵʽΪ_____________��
(3)��֪������ʵĵ���ƽ�ⳣ�����±���
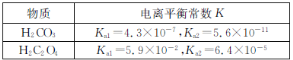
��0.1 mol��L-1��Na2CO3��Һ��pH_______(������������С��������������)0.1 mol��L-1��Na2C2O4��Һ��pH��
��������Ũ�ȵIJ�����Һ��̼����Һ�������ϣ���Һ������Ũ�ȴ�С��˳����ȷ����_________(����ĸ)��
a.c(H+)>c(HC2O4-)>c(HCO3-)>c(CO32-) b.c(HCO3-)>c(HC2O4-)>c(C2O42-)>c(CO32-)
c.c(H+)>c(HC2O4-)>c(CO32-)>c(C2O42-) d.c(H2CO3)>c(HCO3-)>c(HC2O4-)>c(CO32-)