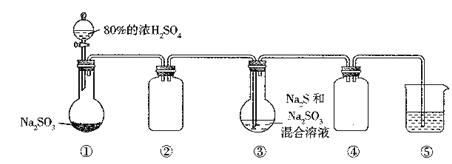
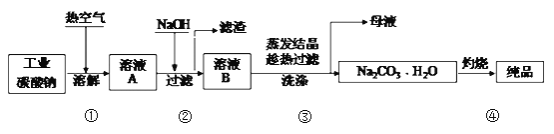
��Ŀ����
����Ŀ����ѧʵ����ѧϰ���⻯ѧ֪ʶ�Ļ���,�����������������ͷḻ���ں��ڻ�ѧѧϰ�з����Ŷ��صĹ��ܺ����á���ش��������⣺
I .ʵ�������ù���NaOH����100 mL 1mol��L-1��NaOH��Һ��
��1������������Һ�����в����õ���������_______________(����Żش�)��
A.�ձ� B.���Թ� C.������ D.100 mL����ƿ
��2�������������⣬��һ��Ҫ�IJ���������______________��
��3����������ƽ��ȡNaOH���������Ϊ_________g�����±���ѡ�����NaOH��������Ҫ������ __________________������ţ���

��4���������ʹ�������Һ��Ũ����α仯�����ƫ�ߡ��� ƫ�͡����䡱��
A.δϴ���ܽ��������Ƶ��ձ�_________��
B.����ƿʹ��ǰ������ˮϴ�����ڱڸ���ˮ���δ���ﴦ��___________��
C.����ʱ��ˮ�����˿̶��ߣ��������Һ������___________��
II����֪��������Ҷ�������ˮ������������������ڻ����98%��Ũ���ᣬ���Ҳ��ܡ�����һ�ݼ��ҵĻ������Ʒ��ͨ��ʵ����з��룬�ɵõ�����ף�ʵ����ʹ�õĹ����������ڹ���ǿ����Һ���������������������������ѧ��Ӧ����
����д���пհף���������������õ������ʵ����ơ�
��� | ʵ�鲽�� | ����ʵ��������������������װʵ��װ�ã� | |
�� | �ܽ� | �����������ձ��У�����98% H2SO4____�� | |
�� | _____________ | ||
�� | ϡ�ͣ������� | ____________ | |
�� | ���� | ||
�� | _________ | ��ܵĹ�������ע����������ˮ��ʹˮ������������ˮ�˳����ٴμ�ˮϴ�ӣ���ϴ���Ρ� | |
�� | ��������Ƿ�ϴ�� | ____________________�� | |
���𰸡� B ��ͷ�ι� 4.0 abe ƫ�� ���� ƫ�� ��ֽ��裬ֱ�����岻���ܽ� ���� ������������Һ���ձ��ڱ�����ע������ˮ�У������Ͻ��裬ֱ������ȫ������ ϴ�ӳ��� ��С�ԹܴӢݵ�©���¿�ȡ����ϴ��Һ������BaCl2��Һ����û�а�ɫ������˵��������ϴ��
��������I .��1�����Թ����ڷ�����ѧ��Ӧ����2������ʱҪ�ý�ͷ�ιܣ���3��m(NaOH)=0.1L��1mol��L-1��40g��mol��1=4.0g�����������Ʒ����ձ�����������ƽ��������4������c=n/V������II������������ֻ����Ҫ��ԭ�е���ɷֿ���������˼·Ӧ�ӡ���֪��������Ҷ�������ˮ�������������������ڻ����98%��������Ҳ��ܡ���������Ȼ��һ�ֿ����������������ڻ����98%�����ᣬ���Ѿ��������Ƕ�����Ũ�����е�״̬��ͬ���ɲ��ù��˵ķ��������߷ֿ�����Ȼ��������ҺΪŨ���ᣬ����ʹ�õIJ�����ͨ����ֽ��ӦΪ����ǿ����Һ��������Թ�����������ˮϴ�ӣ����ɵõ�������Ũ����������Թ��壮��һ�ֹ����ܽ���Ũ�����У�Ҫ�õ�������Ҫ�ٴ��о�����֪��������Ҷ�������ˮ�������������������ڻ����98%�����ᡱ����˵������ϡ�����в��ܽ⣬Ҳ����˵��ֻҪ���Ǽ�ˮϡ�ͼ�Ũ������Һ���Ϳ��ü״���Һ���������������
I .ʵ�������ù���NaOH����100 mL 1mol��L-1��NaOH��Һ����1������������Һ�������õ���������B���Թܣ�A.�ձ������ܽ⣻ C.���������ڽ��衢������D.100 mL����ƿ��������һ���������Һ����ѡB����2�������������⣬����ʱҪ�ý�ͷ�ιܵμ�����Һ�壬�ʻ�һ��Ҫ�IJ��������н�ͷ�ιܡ���3��m(NaOH)=0.1L��1mol��L-1��40g��mol��1=4.0g����������ƽ��ȡNaOH���������Ϊ4.0g�����������Ʒ����ձ�����������ƽ����������NaOH��������Ҫ������ abe������ţ�����4��A.δϴ���ܽ��������Ƶ��ձ����������ʱ��٣���Ũ��ƫ�� ��B.����ƿʹ��ǰ������ˮϴ�����ڱڸ���ˮ���δ���ﴦ��������ʱҪ��ˮ���ʶ�Ũ��û��Ӱ�죬�����䣻C.����ʱ��ˮ�����˿̶��ߣ��������Һ���������������ʱ��٣���Ũ��ƫ�͡�
II�����ܽ⣬�����������ձ��У�����98% H2SO4����ֽ��裬ֱ�����岻���ܽ⡣�ڹ��˷��벻����Ϳ�������ձ��еĻ��Һ�ز�������������������й��ˣ���ϡ�ͣ����������ʽ�����������Һ���ձ��ڱ�����ע������ˮ�У������Ͻ��裬ֱ������ȫ�����壻�ܹ��ˢ�ֱ����������м�������ˮϴ�ӳ������ظ�2-3�Σ���ϴ�ӳ�������ܵĹ�������ע����������ˮ��ʹˮ������������ˮ�˳����ٴμ�ˮϴ�ӣ���ϴ���Ρ���������Ƿ�ϴ������С�ԹܴӢݵ�©���¿�ȡ����ϴ��Һ������BaCl2��Һ����û�а�ɫ������˵��������ϴ����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ����������ʵ�����һ��ʱ�����Һ�о��а�ɫ�������ɣ����н�������ȷ����
ʵ��� | ʵ��� | ʵ��� |
|
|
|
A. ʵ��������ɵij�����BaCO3
B. ʵ���������������
C. ʵ���������п��ܺ���BaSO4
D. ʵ�������ɳ��������ӷ���ʽ�ǣ�Ba2++ H2SO3===BaSO3��+2H+