��Ŀ����
[��ѧ-���ʽṹ������]
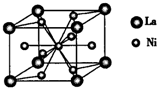
A��B��C��D��E��F����Ԫ�أ�A��B��C���ڵ��������ҵ��ʾ�Ϊ���壮ԭ�Ӵ��ڻ�̬ʱ��A��Bԭ�Ӻ�����Ӿ��ɶԣ��Һ˵����A��B��C��A�����ڱ�����ͬһ����Ԫ�أ���ͼ�ǽ���B��C���γɵ�ij�ֺϽ�ľ����ṹʾ��ͼ��D��E��F��λ��p����Dԭ���ڻ�̬ʱp���������ҵ縺����ͬ��Ԫ�������ģ�E��F��ԭ�Ӿ��������ܲ㣬Eԭ�ӵĵ�һ�����������ĵ����ܣ�kJ/mol���ֱ�Ϊ��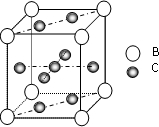
578��1817��2745��11575��E��F���γ�ԭ����Ŀ��Ϊ1��3���۵�Ϊ190��Ļ�����Q����ش�
��1��д��A�Ļ�̬��������Ų�ʽ
��2��C2+��A2+������NH3��H2O���γ���λ��Ϊ4������[A��NH3��4]2+��[C��NH3��4]2+�����жԳƵĿռ乹�ͣ�[C��NH3��4]2+�е�����NH3������Cl-ȡ���ܵõ����ֲ�ͬ�ṹ�IJ����[A��NH3��4]2+�е�����NH3������Cl-ȡ��ֻ�ܵõ�һ�ֲ����[C��NH3��4]2+�Ŀռ乹��Ϊ
��3��1mol D�ĵ��ʷ����д���
��4��ʵ������̬��ˮQ��1.01��105Pa��t1����ܶ�Ϊ5.00g/L������Ħ�����Ϊ53.4L/mol�����ʱQ�����Ϊ��д��ѧʽ��
A��B��C��D��E��F����Ԫ�أ�A��B��C���ڵ��������ҵ��ʾ�Ϊ���壮ԭ�Ӵ��ڻ�̬ʱ��A��Bԭ�Ӻ�����Ӿ��ɶԣ��Һ˵����A��B��C��A�����ڱ�����ͬһ����Ԫ�أ���ͼ�ǽ���B��C���γɵ�ij�ֺϽ�ľ����ṹʾ��ͼ��D��E��F��λ��p����Dԭ���ڻ�̬ʱp���������ҵ縺����ͬ��Ԫ�������ģ�E��F��ԭ�Ӿ��������ܲ㣬Eԭ�ӵĵ�һ�����������ĵ����ܣ�kJ/mol���ֱ�Ϊ��

578��1817��2745��11575��E��F���γ�ԭ����Ŀ��Ϊ1��3���۵�Ϊ190��Ļ�����Q����ش�
��1��д��A�Ļ�̬��������Ų�ʽ
1s22s22p63s23p63d104s2
1s22s22p63s23p63d104s2
�������ʽ��ʾB��C�Ͻ�����CaCu5
CaCu5
��Ԫ�ط��ű�ʾ����2��C2+��A2+������NH3��H2O���γ���λ��Ϊ4������[A��NH3��4]2+��[C��NH3��4]2+�����жԳƵĿռ乹�ͣ�[C��NH3��4]2+�е�����NH3������Cl-ȡ���ܵõ����ֲ�ͬ�ṹ�IJ����[A��NH3��4]2+�е�����NH3������Cl-ȡ��ֻ�ܵõ�һ�ֲ����[C��NH3��4]2+�Ŀռ乹��Ϊ
ƽ��������
ƽ��������
��[A��NH3��4]2+��A���ӻ���ʽΪsp3
sp3
����3��1mol D�ĵ��ʷ����д���
NA
NA
���Ҽ�NA��
NA��
�м���D����̬�⻯���������H+��ϣ�Cԭ����H+���γɵļ�����λ��
���
����4��ʵ������̬��ˮQ��1.01��105Pa��t1����ܶ�Ϊ5.00g/L������Ħ�����Ϊ53.4L/mol�����ʱQ�����Ϊ��д��ѧʽ��
Al2Cl6
Al2Cl6
��Q��������
����
���壮������A��B��C��D��E��F����Ԫ�أ�D��E��F��λ��p����Dԭ���ڻ�̬ʱp���������ҵ縺����ͬ��Ԫ�������ģ���Dԭ�Ӵ��ڵڶ����ڣ���Χ�����Ų�Ϊ2s22p3����DΪ��Ԫ�أ�E��F��ԭ�Ӿ��������ܲ㣬Eԭ�ӵĵ�һ�����������ĵ����ܣ�kJ/mol���ֱ�Ϊ��578��1817��2745��11575��E�����Ϊ+3����EΪ��Ԫ�أ�E��F���γ�ԭ����Ŀ��Ϊ1��3���۵�Ϊ190��Ļ����FΪ-1�ۣ���FΪ��Ԫ�أ�������Q�۵�ͣ�������Q�Ƿ��Ӿ��壻A��B��C���ڵ��������ҵ��ʾ�Ϊ����ԭ�Ӵ��ڻ�̬ʱ��A��Bԭ�Ӻ�����Ӿ��ɶԣ�A��Bԭ����Χ�����Ų�Ϊ4s2��3d104s2��4s24p6���Һ˵����A��B����BΪ��Ԫ�أ�AΪпԪ�أ�C��A�����ڱ�����ͬһ����Ԫ�أ�����ds������CΪͭԪ��
����⣺A��B��C��D��E��F����Ԫ�أ�D��E��F��λ��p����Dԭ���ڻ�̬ʱp���������ҵ縺����ͬ��Ԫ�������ģ���Dԭ�Ӵ��ڵڶ����ڣ���Χ�����Ų�Ϊ2s22p3����DΪ��Ԫ�أ�E��F��ԭ�Ӿ��������ܲ㣬Eԭ�ӵĵ�һ�����������ĵ����ܣ�kJ/mol���ֱ�Ϊ��578��1817��2745��11575��E�����Ϊ+3����EΪ��Ԫ�أ�E��F���γ�ԭ����Ŀ��Ϊ1��3���۵�Ϊ190��Ļ����FΪ-1�ۣ���FΪ��Ԫ�أ�������Q�۵�ͣ�������Q�Ƿ��Ӿ��壻A��B��C���ڵ��������ҵ��ʾ�Ϊ����ԭ�Ӵ��ڻ�̬ʱ��A��Bԭ�Ӻ�����Ӿ��ɶԣ�A��Bԭ����Χ�����Ų�Ϊ4s2��3d104s2��4s24p6���Һ˵����A��B����BΪ��Ԫ�أ�AΪпԪ�أ�C��A�����ڱ�����ͬһ����Ԫ�أ�����ds������CΪͭԪ�أ�
��AΪпԪ�ء�BΪ��Ԫ�ء�CΪͭԪ�ء�DΪ��Ԫ�ء�EΪ��Ԫ�ء�FΪ��Ԫ�أ�
��1��AΪпԪ�أ�A�Ļ�̬��������Ų�ʽΪ1s22s22p63s23p63d104s2���ɾ����ṹ��֪��������Caԭ����ĿΪ8��
=1��Cuԭ����ĿΪ1+8��
=5��B��C�Ͻ�����ΪCaCu5��
�ʴ�Ϊ��1s22s22p63s23p63d104s2��CaCu5��
��2��AΪпԪ�ء�CΪͭԪ�أ�[Zn��NH3��4]2+��[Cu��NH3��4]2+�����жԳƵĿռ乹�ͣ�[Cu��NH3��4]2+�е�����NH3������Cl-ȡ���ܵõ����ֲ�ͬ�ṹ�IJ��[Cu��NH3��4]2+�Ŀռ乹��Ϊƽ�������Σ�[Zn��NH3��4]2+�е�����NH3������Cl-ȡ��ֻ�ܵõ�һ�ֲ��[Zn��NH3��4]2+Ϊ��������ṹ��Zn��ȡsp3�ӻ���
�ʴ�Ϊ��ƽ�������Σ�sp3��
��3��DΪ��Ԫ�أ������е�ԭ��֮���γ�N��N������N��N����Ϊ1���Ҽ���2���м�������1mol�����к���NA�Ҽ���2NA���м���������H+������ɣ��������пչ�������������е�ԭ����1�Թ¶Ե��Ӷԣ�������H+��Ӧ��ͨ����λ������NH4+��
�ʴ�Ϊ��NA��NA����λ����
��4��ʵ������̬��ˮQ��1.01��105Pa��t1����ܶ�Ϊ5.00g/L������Ħ�����Ϊ53.4L/mol���û������Ħ������Ϊ5.00g/L��53.4L/mol=267g/mol�������Q��ѧʽΪ��AlCl3��n�����ԣ�n����27+35.5��3��=267�����n=2�����Ի�����Q��ѧʽΪAl2Cl6��������Q�۵�Ϊ190�棬�۵�ͣ�������Q�Ƿ��Ӿ��壬
�ʴ�Ϊ��Al2Cl6�����ӣ�
��AΪпԪ�ء�BΪ��Ԫ�ء�CΪͭԪ�ء�DΪ��Ԫ�ء�EΪ��Ԫ�ء�FΪ��Ԫ�أ�
��1��AΪпԪ�أ�A�Ļ�̬��������Ų�ʽΪ1s22s22p63s23p63d104s2���ɾ����ṹ��֪��������Caԭ����ĿΪ8��
| 1 |
| 8 |
| 1 |
| 2 |
�ʴ�Ϊ��1s22s22p63s23p63d104s2��CaCu5��
��2��AΪпԪ�ء�CΪͭԪ�أ�[Zn��NH3��4]2+��[Cu��NH3��4]2+�����жԳƵĿռ乹�ͣ�[Cu��NH3��4]2+�е�����NH3������Cl-ȡ���ܵõ����ֲ�ͬ�ṹ�IJ��[Cu��NH3��4]2+�Ŀռ乹��Ϊƽ�������Σ�[Zn��NH3��4]2+�е�����NH3������Cl-ȡ��ֻ�ܵõ�һ�ֲ��[Zn��NH3��4]2+Ϊ��������ṹ��Zn��ȡsp3�ӻ���
�ʴ�Ϊ��ƽ�������Σ�sp3��
��3��DΪ��Ԫ�أ������е�ԭ��֮���γ�N��N������N��N����Ϊ1���Ҽ���2���м�������1mol�����к���NA�Ҽ���2NA���м���������H+������ɣ��������пչ�������������е�ԭ����1�Թ¶Ե��Ӷԣ�������H+��Ӧ��ͨ����λ������NH4+��
�ʴ�Ϊ��NA��NA����λ����
��4��ʵ������̬��ˮQ��1.01��105Pa��t1����ܶ�Ϊ5.00g/L������Ħ�����Ϊ53.4L/mol���û������Ħ������Ϊ5.00g/L��53.4L/mol=267g/mol�������Q��ѧʽΪ��AlCl3��n�����ԣ�n����27+35.5��3��=267�����n=2�����Ի�����Q��ѧʽΪAl2Cl6��������Q�۵�Ϊ190�棬�۵�ͣ�������Q�Ƿ��Ӿ��壬
�ʴ�Ϊ��Al2Cl6�����ӣ�
���������⿼���������Ų����ɡ�ԭ�ӽṹ�����ʹ�ϵ������ṹ�뻯ѧ���ȣ��Ѷ��еȣ����ݺ�������Ų��ƶ�Ԫ���ǹؼ����Ƕ�֪ʶ���ۺϿ��飮

��ϰ��ϵ�д�
 ȫ��������ϵ�д�
ȫ��������ϵ�д� һ��һ����ʱ���ϵ�д�
һ��һ����ʱ���ϵ�д�
�����Ŀ
 ��ƽ��ṹ������
��ƽ��ṹ������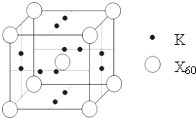 ��2011?����ģ�⣩����ѧ--���ʽṹ�����ʡ�
��2011?����ģ�⣩����ѧ--���ʽṹ�����ʡ�