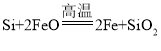��Ŀ����
����Ŀ����ͭ���ǹ�ҵ��ͭ����Ҫԭ�ϣ�����Ҫ�ɷ�ΪCuFeS2������һ����Ȼ��ͭ��������ʯ����Ϊ�˲ⶨ�û�ͭ��Ĵ��ȣ�ijͬѧ���������ʵ�飺
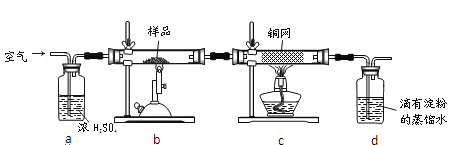
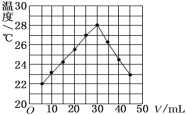
�ֳ�ȡ��ϸ�Ļ�ͭ����Ʒ1.150g���ڿ��������½������գ�����Cu��Fe3O4��SO2���壬ʵ���ȡd����Һ��1/10������ƿ�У���0.05mol/L������Һ���еζ���������Ϊ0.00mL��ĩ������ͼ1��ʾ�����������գ�
��1��������Ʒ���õ�����Ϊ___������Ʒ��ϸ���ٷ�Ӧ����Ŀ����__________��
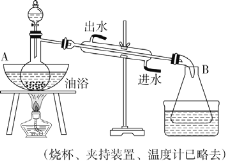
��2��װ��a��������___________��������Ӧ����������ͨһ��ʱ��Ŀ�������Ŀ����___��
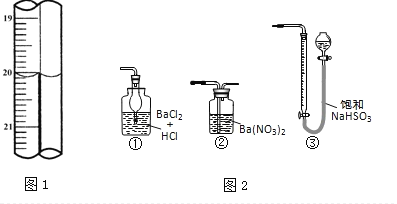
��3���ζ�ʱ��������Һ�������Ϊ___mL���жϵζ��Ѵ��յ��������_____________��
��4��ͨ�������֪���û�ͭ��Ĵ���Ϊ_________��
��5������ͼ2װ���������ʵ��װ��d��ͬ�����Դﵽʵ��Ŀ�ĵ���________��
���𰸡�������ƽ ��߷�Ӧ���ʣ���ʹ��ͭ���ַ�Ӧ ��ȥ�����е�ˮ���������ڹ۲�������� ��װ���еĶ�����������ϳ�װ�ò�ȫ������ 20.00 ��Һ����ɫ�����ɫ����������ڲ���ɫ 80�� ��
��������
��1�����ݳ�����ͭ����Ʒ1.150g��ѡ��ȷ�Ƚϸߵ�����������Ʒ��ϸ�������˽Ӵ������
��2��Ũ������Խ�ˮ��ȥ�������Ը���ð�����ݵ����������ڿ������٣�
(3)��Ӧ�����Ķ�������Ӧ�þ����ܵı�dװ�����գ�
��4�����ݵζ��ܵĶ��������������ĵ���Һ����������ݷ�Ӧ����ʱ����ɫ�仯�жϵζ��յ㣻
��5�����ҳ���ͭ��Ͷ������ⵥ�ʵĹ�ϵʽCuFeS2��2SO2��2I2���ٸ����������ݽ��м��㣻
��6��ͼ2�еĢ���ͨ���������Ӧ���������ᱵ���������Ը������ᱵ����������������������
��1�����ڳ�����ͭ����Ʒ1.150g����ȷ�ȴﵽ��ǧ��֮һ��Ӧ��ѡ�õ�����ƽ���г������ѻ�ͭ����Ʒ��ϸ����������Ӵ�������Ӷ���߷�Ӧ���ʣ�����ʹ��ͭ���ַ�Ӧ��
�ʴ��ǣ�������ƽ����߷�Ӧ���ʣ���ʹ��ͭ���ַ�Ӧ��
��2��װ��a�е�Ũ����������տ����е�ˮ��������ֹˮ�������뷴Ӧװ��b�з���Σ�գ�ͬʱ����ð�������ݵĿ��������������ͨ������
�ʴ�Ϊ����ȥ�����е�ˮ���������ڹ۲�������٣�
(3)��ͭ�����ȷֽ����ɶ��������һϵ�в���ֽ���Ϻ���Ȼ��Ҫͨ��һ��ʱ��Ŀ��������Խ�b��dװ���еĶ�������ȫ���ų�ȥ��ʹ������Ӿ�ȷ��
�ʴ�Ϊ����װ���еĶ����������岢ȫ�����գ�
��4�����ݵζ��ܵ�ʾ�����Ϸ�С���·����Զ����ζ���ʾ����20.00mL�����ﵽ�ζ��յ�ʱ�����������Ѿ����ⵥ��������ϣ��ٵ���һ�εⵥ�ʣ��������ۻ������
�ʴ�Ϊ��20.00����Һ����ɫ�����ɫ����������ڲ���ɫ��
��5��������ԭ���غ�͵����غ��ҳ���ϵʽ��CuFeS22SO22I2�����ĵ�0.05mol/L������Һ20.00mLʱ�������ĵĵⵥ�ʵ���Ϊ��0.05mol/L��0.02L=0.0010 mol�����Ի�ͭ��������ǣ�0.5��0.0010mol��184g/mol��10=0.92g�������䴿���ǣ�![]() ��100%=80%��
��100%=80%��
�ʴ�Ϊ��80%��
��6��ͼ2�У�
�ٲ������ɳ�����������������
�����ᱵ��Һ��ͨ����������ܹ��������ᱵ���������˸���������ᱵ����������������������������ȷ��
���б���������������Һֻ���ų��������������ն������������dװ�ã�����
�ʴ�Ϊ���ڡ�