��Ŀ����
������;ʮ�ֹ㷺���н�����ά���ء�֮�ơ�ij����Ϊ�˴Ӻ������ʵ�VOSO4��Ʒ�л��յõ�����V2O5������������£�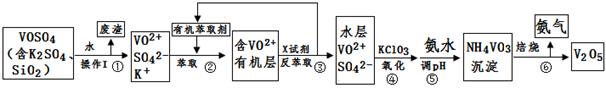
��ش��������⣺
��1����������÷����ijɷ��� ��д��ѧʽ����NH4VO3�ڱ���ǰҪϴ��2-3�Σ��ò���������Ϊ ��
��2������ڡ��۵ı仯���̿ɼ�Ϊ����ʽR��ʾVO2+��HA��ʾ�л���ȡ������
R2(SO4)n (ˮ��)+ 2nHA���л��㣩 2RAn���л��㣩 + nH2SO4 (ˮ��)
2RAn���л��㣩 + nH2SO4 (ˮ��)
���������ȡʱ��������������ԭ���� ��
�������X�Լ�Ϊ ��
��3���ݵ����ӷ���ʽΪ ��
��4���ù��������У�����ѭ�����õ������� �� ��
��5����ȡVOSO4��Ʒwg���������a mol��L��1�������ҺVmL������ػ�ԭ����Ϊ�Ȼ��أ�����1kg��Ʒ�����Ͽ��Ƶõ�V2O5������ g���ú�w��V�Ĵ���ʽ��ʾ����
��1��SiO2��1�֣�����©���м�ˮ����û��������ˮ�������ظ�������2�֣�
��2��������к����ᣬ��ʹƽ�������ƶ�����߷�����ȡ��(�����Ʊ����������RAn���л��㣩��Ũ�ȡ��ٷֺ�����)��2�֣��� H2SO4��1�֣�
��3��NH3��H2O+VO3�� =NH4VO3��+OH����3�֣�
��5����������ˮ�����л���ȡ����2�֣���
��6��546av/w �� ��3�֣�
����������������ݹ�������ͼ֪����������1���ܽ�õ��ķ�����SiO2�� ������ڣ�2��С�ⷴӦ֪������2��3�е���ȡ�ͷ���ȡ��������ƽ���ƶ�ԭ��ʵ����R2(SO4)n���л����ˮ���е��ת����ʵ�����ǶԱ��ᴿ��ĸ������̣�������4��5ǰ��ıȽ�֪������4�ǽ�VO2+��������VO3-������5�ֽ�VO3-ת��ΪNH4VO3�����������6�õ�Ŀ�����V2O5��
���㣺�����Թ�������Ϊ��������Ԫ�ؼ��������ѧʵ�������������ѧ����ʽ����д�Լ���ѧ��������֪ʶ��

 ������ʱͬ����ϰ��ϵ�д�
������ʱͬ����ϰ��ϵ�д� ѧҵ����һ��һ��ϵ�д�
ѧҵ����һ��һ��ϵ�д���14�֣�ij�о���ѧϰС���������ռ���������Ϣ�����ƣ��׳Ƴ����һ��ǿ��ԭ�����ڹ�ҵ����;�㷺�����Ƕ�������������Ƶķ�Ӧ����̽����
��ʵ�顿
�ڴ���������Һ�м���������Һ��������Һʼ�ճ��塣
��1�������й����Ƶ�˵����ȷ���� ��
a. ������Һ���˾��ã�Ӧ��������
b. ������Һ��ͨ��SO2�е���ɫ���ǣ���֤��SO2��������
c. �����ڿ����г��Ⲣ���Ϸų�H2S���壬˵�����ԣ�H2CO3>H2S
d. ����������Һʱ����������������
e. ��������Һ�еμ�ϡ�����г�������ζ����������
��������衿
��2����Ӧ�����Һ�п϶�����Cl����SO42����OH����Ϊ̽����Һ�п��ܻ����ڵ����������ӣ�����������ˮ�⼰������CO2��Ӱ�죩����ѧϰС��������¼��裺
����1��ֻ������S2��
����2��ֻ������ClO��
����3��
�����ʵ�鷽������֤���衿
��3����д��ʵ�鲽���Լ�Ԥ������ͽ��ۡ�
��ѡʵ���Լ���1 mol? L��1��CuSO4��Һ��1 mol? L��1NaOH��Һ�����ۣ�KI��Һ����ɫʯ����Һ����̪��Һ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ������Ӧ����Һ���Թ��У��μ� | �� ������� ������ |
| ����:2����ȡ������Ӧ����Һ���Թ��У��μ� | �� ������� ������ �� �� �����3������ |
��˼���뽻����
��4��ʵ��ʱijͬѧ��������������Һ�ʻ�ɫ�����뵽��ҵ����ʻ�ɫ��ԭ��ͬѧ��Ϊ����������Һ�л�������FeCl3�����Ƿ�ͬ���ͬѧ�ĵĹ۵� ���ͬ�⡱��ͬ�⡱������������ɣ� ��
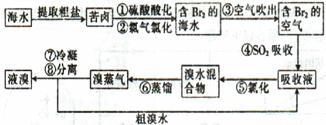
����ֲ���纣���������к��зḻ�ĵ�Ԫ�أ�����Ҫ�Ե⻯����ʽ���ڡ���һ��ѧ����С���ú���Ϊԭ����ȡ�����ⵥ�ʣ����ǽ��������ճɻң���ˮ����һ��ʱ�䣨���õ⻯�����ܽ���ˮ�У����õ�����������Һ��Ȼ������ʵ��������ȡ���ʵ⣺
��1��ָ����ȡ��Ĺ������йص�ʵ��������ƣ��� ���� ��
��2�������������õ��л��Լ������� ��ֻ��һ�֣�������ѡ�������� ��
��3���������̢ۿ��Էֽ�Ϊ���¼�����
| A����ʢ����Һ�ķ�Һ©����������̨����Ȧ�У� |
| B����50ml��ˮ��15ml�л��ܼ�������ѡ�ģ������Һ©���У����Ǻò������� |
| C�������Һ©���������ϿڵIJ������Ƿ�©Һ�� |
| D����ת©������������ʱ�����������������رջ������ѷ�Һ©�������� |
F���ӷ�Һ©���Ͽڵ����ϲ�Һ�壻
G����©���ϿڵIJ�������ʹ���ϵİ�����©���Ͽڵ�С����
H�����á��ֲ㡣
��a�����̢���ȷ���������˳���ǣ���������ĸ�� ��
��b������G��������Ŀ���ǣ� ��
���к͵ζ��ķ����ⶨNaOH��Na2CO3�Ļ����Һ��NaOH�ĺ����������ڻ��Һ�м��������BaCl2��Һ��ʹNa2CO3��ȫת���BaCO3������Ȼ���ñ�����ζ�(��֪�������ָʾ����ɫ��pH��Χ���ټ���3.1��4.4 �ڼ���4.4��6.2 �۷�̪8.2��10)����ش�
��1�������BaCO3������NaOH��Һ�е������ᣬӦѡ�� ָʾ�����жϵ���ζ��յ��ʵ�������� ��
��2��Ϊ�ⶨij�ռ���Ʒ��NaOH�ĺ���(����Ʒ������ΪNa2CO3)��ijͬѧ��������ʵ�飺ȷ��ȡ5.0g��Ʒ���Ƴ�250mL��Һ��Ȼ������θ�ȡ���ƺõ��ռ���Һ20.00mL������������ˮϴ������ƿ�У��ֱ���������BaCl2��Һ��������ƿ�и�����1��2��ָʾ������Ũ��Ϊ0.2000mol��L-1�������Һ���еζ���������ݼ�¼���£�
| ʵ���� | V(�ռ���Һ)/mL | V(HCl)/mL | |
| ������ | ĩ���� | ||
| 1 | 20.00 | 0.00 | 31.00 |
| 2 | 20.00 | 1.00 | 32.04 |
| 3 | 20.00 | 1.10 | 32.18 |
�ڵζ�ʱ����ȷ������ ��
�����в����ᵼ���ռ���Ʒ��NaOH�����ⶨֵƫ�ߵ���
A����ƿ������ˮϴ��δ�ô���Һ��ϴ B����ʽ�ζ���������ˮϴ��δ�ñ�Һ��ϴ
C���ڵζ�ǰ�����ݣ��ζ���������ʧ D���ζ�ǰƽ�Ӷ������ζ��������Ӷ���
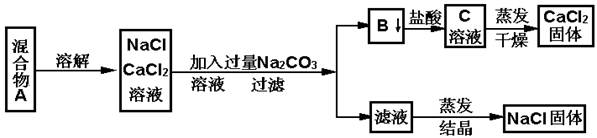

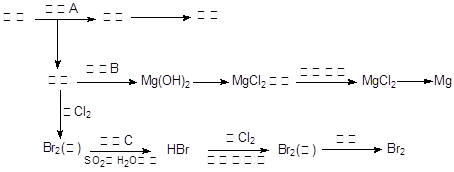

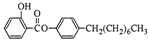 �����£�
�����£�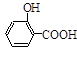 ��ˮ���ᣩ��SOCl2��
��ˮ���ᣩ��SOCl2��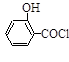 ��ˮ�����ȣ���HCl����SO2��
��ˮ�����ȣ���HCl����SO2��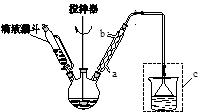
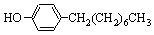 �����¶ȿ�����100�����ң����Ͻ��衣
�����¶ȿ�����100�����ң����Ͻ��衣