��Ŀ����
3����֪����ϩ��һ����ɫ������ζ���壬������ˮ�����������Ȼ�̼���л��ܼ���SO2������ˮ�����������Ȼ�̼���л��ܼ���ʵ��������ϩʱ��������������ʹ��ˮ��ɫ���ס���ͬѧ̽����ˮ��ɫ��ԭ���������Ѽ��飬���ּг�װ���ԣ���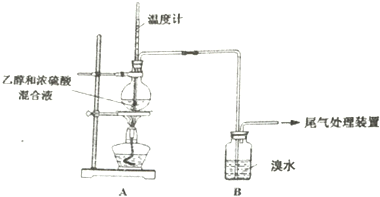
| ���� | ���� |
| ��ȼ�ƾ��ƣ� ������170�� | a��A����ƿ��Һ�彥����� b��B����������ð������Һ����ɫ |
| �� | |
| ʵ����� | c��A����ƿ�ڸ���������ɫ����״��д̼�����ζ�ݳ� |
��2������ʹB����Һ��ɫ�����ʣ�����Ϊ��C2H4������Ϊ�����ų�SO2�����ã�
�ٸ��ݼĹ۵㣬C2H4ʹB����Һ��ɫ�Ļ�ѧ����ʽΪCH2=CH2+Br2��CH2Br-CH2Br��
���Ҹ�������c��Ϊ������SO2��SO2ʹB����Һ��ɫ�Ļ�ѧ����ʽ��SO2+Br2+2H2O=2HBr+H2SO4��
��3��Ϊ֤ʵ���Թ۵㣬�ס���ͬѧ����ʵ�飬������A��B������һ��װ��ij���Լ���ϴ��ƿ�����۲쵽��ˮ��ɫ��
�ٸ��ݼ���ƣ�ϴ��ƿ��ʢ�ŵ��Լ���NaOH��Һ��
����Ϊ��һ����֤��۵㣬ȡ������Ӧ���B����Һ����������BaCl2��Һ��������������ɫ��������Ӧ�����ӷ���ʽΪSO2+2H2O+Br2�T4H++2Br-+SO42-��SO42-+Ba2+�TBaSO4����SO2+2H2O+Br2+Ba2+�T4H++2Br-+BaSO4����
���� ��1��ʵ��������ϩ���Ҵ���ŨH2SO4������170������ϩ���Ҵ�������ȥ��Ӧ������̼���������ǻ�������̼�����ڵ�̼�ϵ�̼�����Ũ���������ˮ�ԡ���ˮ�Ժ�ǿ�����ԣ��������Ҵ���Ũ���ᱻ��ԭ������SO2��ͬʱ���Ҵ��е��������ˮ�������ȥ��
��2������ϩ�еĹ�����Ϊ˫�����ܹ�����ӳɷ�Ӧ����ĺ���ɫ��ȥ��
���Ҹ�������c��Ϊ������SO2���嵥�ʿ��ԺͶ���������������ԭ��Ӧ����������������
��3�����������ƿ��ԺͶ�����������Ӧ�����ն��������ų���������ĸ��ţ�����������������Ʒ�Ӧ�����������ƺ�ˮ��
���嵥�ʿ��ԺͶ���������������ԭ��Ӧ����������Ӻͱ����ӷ�Ӧ�����ɰ�ɫ������
��� �⣺��1��ʵ��������ϩ���Ҵ���ŨH2SO4���ȷ�����ȥ��Ӧ������ϩ��ԭ������ʽΪ��CH3-CH2-OH $��_{170��}^{Ũ����}$CH2=CH2��+H2O��Ũ���������ˮ�ԣ���ʹ�Ҵ��е��������ˮ�������ȥ����Һ��������ڡ�����ӦΪ��CH3CH2OH+4H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$4SO2��+CO2��+7H2O+C��ͬʱŨ�������ǿ�����ԣ�
�ʴ�Ϊ��CH3-CH2-OH $��_{170��}^{Ũ����}$CH2=CH2��+H2O����ˮ�ԡ�ǿ������
��2���ٸ��ݼĹ۵㣬��ˮ����ʹB����Һ��ɫ��˫���䵥�����ϼ������ϼӳ��Լ�ԭ����ԭ�ӣ���Ӧ�Ļ�ѧ����ʽΪ��CH2=CH2+Br2��CH2Br-CH2Br��
�ʴ�Ϊ��CH2=CH2+Br2��CH2Br-CH2Br��
���Ҹ�������c��Ϊ������SO2������������л�ԭ�ԣ��嵥�ʾ��������ԣ���B���嵥�ʿ��ԺͶ���������������ԭ��Ӧ����Ӧ�Ļ�ѧ����ʽΪ��SO2+Br2+2H2O=2HBr+H2SO4��
�ʴ�Ϊ��SO2+Br2+2H2O=2HBr+H2SO4��
��3���ٸ��ݼ���ƣ�ϴ��ƿ��ʢ�ŵ��Լ����������ƣ����ԺͶ�������Ӧ�����ն��������ų���������ĸ��ţ���ѧ����ʽΪ��SO2+2NaOH=Na2SO3+H2O��
�ʴ�Ϊ��NaOH��Һ��
���嵥�ʿ��ԺͶ���������������ԭ��Ӧ��������������Ӻ������ӣ���������Ӻͱ����ӷ�Ӧ�����ɰ�ɫ��������ط�Ӧ�����ӷ���ʽΪSO2+2H2O+Br2�T4H++2Br-+SO42-��SO42-+Ba2+�TBaSO4����SO2+2H2O+Br2+Ba2+�T4H++2Br-+BaSO4����
�ʴ�Ϊ��SO2+2H2O+Br2�T4H++2Br-+SO42-��SO42-+Ba2+�TBaSO4����SO2+2H2O+Br2+Ba2+�T4H++2Br-+BaSO4����
���� ���⿼��ѧ���Ҵ��Ļ�ѧ���ʣ���ֿ���ѧ���ķ������������Ӧ�û���֪ʶ�����������շ�Ӧԭ������ȡ�����Ϣ����ȷ��д����ʽ�ǽ��ؼ�����Ŀ�Ѷ��еȣ�

 ��ǰ����ϵ�д�
��ǰ����ϵ�д�| A�� | K+��Cl-��MnO4-��SO42- | B�� | Na+��K+��AlO2-��NO3- | ||
| C�� | Na+��NO3-��SO42-��HCO3- | D�� | Na+��Cu2+��NO3-��CO32- |
| װ�� | ʵ����� | �Թ��е�ҩƷ | ���� |
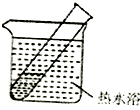 | ʵ��� | 2mL������Һ�����ν�ŨNaOH��Һ | �����ݲ�����һ��ʱ�����Һ��ڣ��Թܱڸ������� |
| ʵ��� | 2mL������Һ�����ν�Ũ��ˮ | �����ݲ�����һ��ʱ�����Һ�����Ա仯 |
a��Ag��NH3��2++2H2O?Ag++2NH3H2O
b��AgOH���ȶ������ֽ�Ϊ��ɫAg2O
��1������������Һ�����ҩƷ��2%��������2%ϡ��ˮ��
��2�������飬ʵ������������NH3����ɫ��������Ag2O��
����ʪ��ĺ�ɫʯ����ֽ����NH3�����������Ǻ�ɫʯ����ֽ������
�ڲ���Ag2O��ԭ����ˮԡ���ȣ���ʹNH3•H2O�ֽ�ɰ����ݳ���ʹ�ÿ��淴Ӧƽ�����ƣ��ٽ��������������������ӵ�ת���������Ӻ����������ӷ�Ӧ��������������AgOH���ȶ���ˮԡ���ȷֽ�Ϊ��ɫAg2O��
��3����ͬѧ�Բ���������ԭ��������裺������NaOH��ԭAg2O��ʵ�鼰������AgNO3��Һ�м������NaOH��Һ�����ֺ�ɫ������ˮԡ���ȣ�δ����������
��4�����¼��裺��NaOH�����¿�����NH3��ԭAg2O������ͼ1��ʾװ�ý���ʵ�飬�����������������߿��ڻ�������ʯ�Һ�Ũ��ˮ��ȡNH3��װ�ü�ͼ���г������ԣ���
��5����ͬѧ��Ϊ�ڣ�4����ʵ���л���Ag��NH3��2OH���ɣ��ɴ���������裺��NaOH�����£�������Ag��NH3��2OHҲ������NH3��ԭAg2O�ķ�Ӧ��������ͼ2ʵ�飺
���в���Ag2O�ܽ��ڰ�ˮ�У��÷�Ӧ�Ļ�ѧ����ʽ��Ag2O+4NH3•H2O=2Ag��NH3��2OH+3H2O��
��ʵ����֤ʵ������������ݵ�����������Һ�Ӵ����Թܱ�������������
����HNO3��ϴ�Թܱ��ϵ�Ag���÷�Ӧ�Ļ�ѧ����ʽ��2HNO3��Ũ��+Ag�TAgNO3+2NO2��+H2O��
| A�� | �����������ڹ����·�Ӧֻ����һ������ | |
| B�� | ���ں��������´���������ɻ����� | |
| C�� | ��ϩ��������Ȼ�̼��Һ��Ӧ����1��2-�������� | |
| D�� | ����Ũ���ᡢŨ���Ṳ�Ȳ�����50-60�淴Ӧ���������� |
| ���Ӵ��� | a | b | c | d | e | f | g |
| ԭ�Ӻ��� | ���� | ���� | ˫�� | ��� | ���� | ��� | ��� |
| ���������λ��ɣ� | 0 | 1+ | 1- | 0 | 2+ | 1+ | 0 |
�����
��1��a���ӵ�ԭ�ӽṹʾ��ͼ
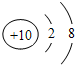 ��b���ӵĵ����Ų�ʽ1s22s22p6��
��b���ӵĵ����Ų�ʽ1s22s22p6����2��b��e��ӦԪ�ص�����������Ӧˮ����ļ���ǿ���Ƚ�ΪNaOH��Mg��OH��2���û�ѧʽ��ʾ����
��3����d��������ˮ����ˮ��Һ�еķ�Ӧ����ʽΪNH3+H2O=NH3•H2O��d���ӵĿռ乹��Ϊ�����Σ�����Ϊ107.5�㣻��dΪ����������ӣ������ʽΪ
 ��
����4��c������OH-��f������H3O+���û�ѧʽ��ʾ������c��f����g�����ӷ���ʽΪOH-+H3O+=2H2O��
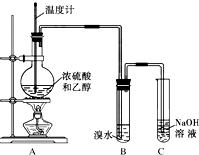 Ϊ̽����ϩ����ȡ����ϩ����ˮ��Ӧ���й����⣬�ס��ҡ�����ͬѧ����������ʵ��������������ǵĻ���ش�������⣮
Ϊ̽����ϩ����ȡ����ϩ����ˮ��Ӧ���й����⣬�ס��ҡ�����ͬѧ����������ʵ��������������ǵĻ���ش�������⣮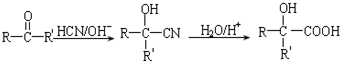
 ��
�� ��
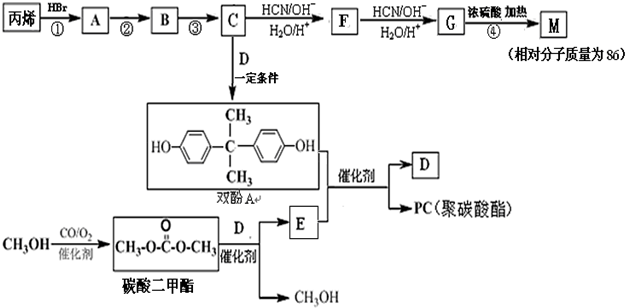
��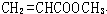 ��
�� ��
��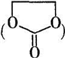 ����CH3OH������������Ӧ����̼�����������һ�ִ���д���÷�Ӧ�Ļ�ѧ����ʽ
����CH3OH������������Ӧ����̼�����������һ�ִ���д���÷�Ӧ�Ļ�ѧ����ʽ
 ��
�� ��
��
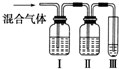 Ϊ̽����ϩ����ļӳɷ�Ӧ����ͬѧ��Ʋ�����������ʵ�飺�����Ҵ���Ũ����Ϊԭ����ȡ��ϩ�������ɵ�����ֱ��ͨ����ˮ�У�������Һ��ɫ����֤����ϩ����ˮ�����˼ӳɷ�Ӧ��
Ϊ̽����ϩ����ļӳɷ�Ӧ����ͬѧ��Ʋ�����������ʵ�飺�����Ҵ���Ũ����Ϊԭ����ȡ��ϩ�������ɵ�����ֱ��ͨ����ˮ�У�������Һ��ɫ����֤����ϩ����ˮ�����˼ӳɷ�Ӧ��