��Ŀ����
��16�֣�ҽ���Ȼ��ƿ����ڲ��ƿ������ȣ��Թ�ҵ̼��ƣ�������Na+��Al3+��Fe3+�����ʣ�����ҽ�ö�ˮ���Ȼ��ƣ�CaCl2 ��2H2O����������Ϊ97.3��103.0%������������Ϊ��
(�����õ���ԭ������Cl 35.5 Ca 40 O 16 )
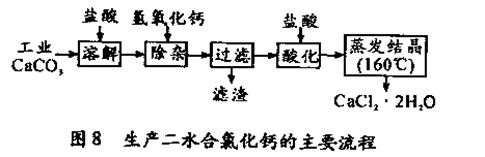
��1��CaCO3�����ᷴӦ�����ӷ���ʽ ��
��2������ʱ���õIJ����������ձ������������ ��������Ҫ�ɷֵĻ�ѧʽ ��
��3���ữʱ�������Ŀ��Ϊ��
�� ��
�ڷ�ֹCa2+ ������ʱ����ˮ�⡣
��4��Ϊʲô�����ᾧҪ������160�棺 ��
��
��5��Ϊ�ⶨ��Ʒ��CaCl2 ��2H2O�ĺ�������ȡ0.7522g��Ʒ�����250mL��Һ���ֱ�ȡ����Һ25.00mL��������ƿ�У���0.04mol/LAgNO3��Һ�������εζ������� AgNO3��Һ��ƽ�����Ϊ20.39mL��
��ͨ�����㣬��Ʒ�к�CaCl2 ��2H2O���������� ����ֻд����ʽ������������
����������Ʒ CaCl2 ��2H2O����������ƫ�ߣ���������ʵ���������ܵ�ԭ��֮һΪ ��
��
(�����õ���ԭ������Cl 35.5 Ca 40 O 16 )

��1��CaCO3�����ᷴӦ�����ӷ���ʽ ��
��2������ʱ���õIJ����������ձ������������ ��������Ҫ�ɷֵĻ�ѧʽ ��
��3���ữʱ�������Ŀ��Ϊ��
�� ��
�ڷ�ֹCa2+ ������ʱ����ˮ�⡣
��4��Ϊʲô�����ᾧҪ������160�棺
 ��
����5��Ϊ�ⶨ��Ʒ��CaCl2 ��2H2O�ĺ�������ȡ0.7522g��Ʒ�����250mL��Һ���ֱ�ȡ����Һ25.00mL��������ƿ�У���0.04mol/LAgNO3��Һ�������εζ������� AgNO3��Һ��ƽ�����Ϊ20.39mL��
��ͨ�����㣬��Ʒ�к�CaCl2 ��2H2O���������� ����ֻд����ʽ������������
����������Ʒ CaCl2 ��2H2O����������ƫ�ߣ���������ʵ���������ܵ�ԭ��֮һΪ
 ��
����16�֣�
(1) CaCO3+ 2H+ ==Ca2+ + CO2��+ H2O ��3�֣�
(2) ��ͨ©����2�֣� ��Fe(OH)3�� Al(OH)3��2�֣�
��3����ȥ�������������ƣ�2�֣���������֣�����4���¶�̫��CaCl2 ��2H2O��ʧˮ��2�֣�
��5��(2��147��20.39��10-4/0.7522 )��100%(3��) ��Na+û��ȥ����NaCl,���� �ⶨ���ƫ��2�֣���������֣�
�ⶨ���ƫ��2�֣���������֣�
(1) CaCO3+ 2H+ ==Ca2+ + CO2��+ H2O ��3�֣�
(2) ��ͨ©����2�֣� ��Fe(OH)3�� Al(OH)3��2�֣�
��3����ȥ�������������ƣ�2�֣���������֣�����4���¶�̫��CaCl2 ��2H2O��ʧˮ��2�֣�
��5��(2��147��20.39��10-4/0.7522 )��100%(3��) ��Na+û��ȥ����NaCl,����
 �ⶨ���ƫ��2�֣���������֣�
�ⶨ���ƫ��2�֣���������֣���

��ϰ��ϵ�д�
�����Ŀ





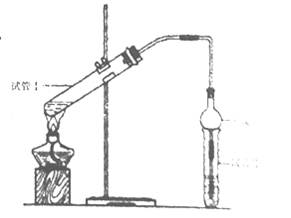
 1)��ͼ����KMnO4��Ũ���ᷴӦ��ȡ���������ļ���װ�á�
1)��ͼ����KMnO4��Ũ���ᷴӦ��ȡ���������ļ���װ�á�