��Ŀ����
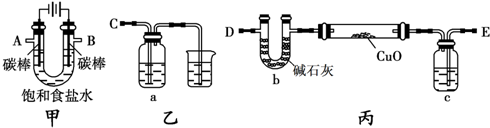
9�������״���C6H5��3C-OH��һ����Ҫ�Ļ���ԭ�Ϻ�ҽҩ�м��壮ʵ���Һϳ������״���ʵ��װ����ͼ1��ʾ����֪���ٹ��������ɵ��м����ʸ����Լ�����ˮ�ⷴӦ��
�ڲ���������ʵķе����£�
| ���� | �е�/�� |
| �����״� | 380 |
| ���� | 34.6 |
| �屽 | 156.2 |
��ش��������⣺
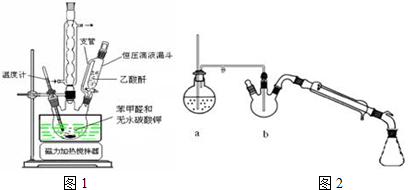
��1��װ���в�������B������Ϊ�����ܣ�װ����ˮCaCl2������A�������Ƿ�ֹ�����е�ˮ��������װ�ã���������Լ�ˮ�⣮
��2��װ���еμ�Һ��δ����ͨ��Һ©�����õ�Һ©����������ƽ��ѹǿ��ʹ©����Һ��˳�����£�
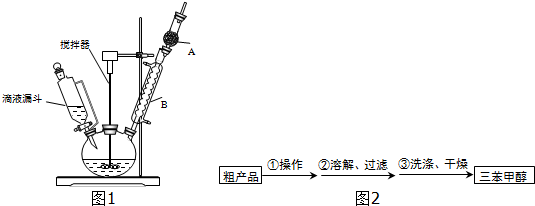
��3���Ƶõ������״��ֲ�Ʒ�к������ѡ��屽���Ȼ�淋����ʣ����������ͼ2�ᴿ���������У������ٵ���������������ϴ��Һ���ѡ��a������ĸ��ţ���
a��ˮ b������ c���Ҵ� d����
�����Ʒ�Ѿ�ϴ�Ӹɾ��IJ���Ϊȡ�������һ��ϴ��Һ���Թ��У��μ���������Һ�����������ɣ����Ѿ�ϴ�Ӹɾ�����֮��δϴ�Ӹɾ���
��4�����Ȳⶨ����ȡ2.60g��Ʒ�����������Һ���������������ƣ��������Ʋ���Ӧ������ַ�Ӧ������ɵ������ڱ�״���µ����Ϊ100.80mL�����Ʒ�������״�����������Ϊ90%��
���� ��1��ͼ�в�������B�������������ܣ����ڸ����Լ�����ˮ�⣬B�������Ƿ�ֹ�����е�ˮ��������װ�ã�
��2��װ���еμ�Һ��δ����ͨ��Һ©�����õ�Һ©����������ƽ��ѹǿ��
��3�������״��ֲ�Ʒ�к������ѡ��屽���������������л�������״��ķе���ߣ�������������ķ�����ȥ�л����ʣ���ʽ�廯þ����ˮ���������л��ܼ�������ϴ��Һѡ��ˮ�����ϴ���Ƿ�ɾ���ȡ�������һ��ϴ��Һ���Թ��У�����������Һ�����Ƿ��������ӣ�
��4����2-OH��H2�ɼ���������״������ʵ������ٸ���m=nM���������״������������������Ʒ�������״�������������
��� �⣺��1��ͼ�в�������B�������������ܣ����ڸ����Լ�����ˮ�⣬B�������Ƿ�ֹ�����е�ˮ��������װ�ã������ʽ�Լ�ˮ�⣬
�ʴ�Ϊ�������ܣ���ֹ�����е�ˮ��������װ�ã���������Լ�ˮ�⣻
��2��װ���еμ�Һ��δ����ͨ��Һ©�����õ�Һ©����������ƽ��ѹǿ��ʹ©����Һ��˳�����£�
�ʴ�Ϊ��ƽ��ѹǿ��ʹ©����Һ��˳�����£�
��3�������״��ֲ�Ʒ�к������ѡ��屽���������������л�������״��ķе���ߣ�����������������ķ�����ȥ�л����ʣ�
���ڼ�ʽ�廯þ����ˮ���������л��ܼ�������ϴ��Һѡ��ˮ����ѡa��
���ϴ���Ƿ�ɾ���һ�㲽���ǣ�ȡ�������һ��ϴ��Һ���Թ��У��μ���������Һ�����������ɣ�����ϴ�Ӹɾ�����֮��δϴ�Ӹɾ���
�ʴ�Ϊ����������a��ȡ�������һ��ϴ��Һ���Թ��У��μ���������Һ�����������ɣ�����ϴ�Ӹɾ�����֮��δϴ�Ӹɾ���
��4����2-OH��H2����֪�����״������ʵ�����$\frac{0.1008L}{22.4L/mol}$��2=0.009mol�����Բ�Ʒ�������״���������0.009mol��260g/mol=2.34g�����Ʒ�������״�����������=$\frac{2.34g}{2.60g}$��100%=90%��
�ʴ�Ϊ��90%��
���� ���⿼���л���ϳ�ʵ�顢���ʵķ�����ϴ�ӵȻ�����������ʵ��װ�õķ������۵ȣ��ϺõĿ���ѧ�������ݵ�Ӧ�á��Ķ���ȡ��Ϣ�������Լ�֪ʶǨ��Ӧ�ã��Ѷ��еȣ�

| A�� | �٢ڢۢܢ� | B�� | �ڢۢܢݢ� | C�� | �ݢڢ٢ۢ� | D�� | �ܢ٢ڢݢ� |
| �ܽ��ԣ������������ܼ��� | �е㣨�棩 | �ܶȣ�g/mL�� | |
| �Ҵ� | ��ˮ���ܣ��������л��ܼ� | 78.5 | 0.8 |
| ������ | ������ˮ���������л��ܼ� | 38.4 | 1.4 |
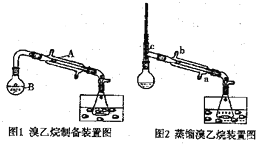
��1��������A�������ǣ�ֱ�������ܣ�
������B���ݻ��ǣ�c��
a��150mLb��200mLc��250mL
��2������ˮ���е�Һ����DZ�ˮ��ʹ�ñ�ˮ��Ŀ���ǽ��£�����������Ļӷ���
��3��HBr���Ҵ���Ӧ����Ŀ�����Ļ�ѧ����ʽHBr+CH3CH2OH$\stackrel{��}{��}$CH3CH2Br+H2O��
��4��ͼ1���ռ����IJ�Ʒ�к���һ����������Br2��Ϊ��ȥ�����ʣ���������Һ�м��������ģ������������Լ���ѡȡ��bd��Ȼ���ٽ��з�Һ�������ɣ�
a�����Ȼ�̼ b��̼����ϡ��Һ c����ˮCaCl2 d����ˮMgSO4
��5��������4���������Ŀ������л�����������ˮ�����ʣ���ͨ�����Ʒ�м���һ������c���������a��d��ѡȡ����������ú��ˣ�����Һʹ��ͼ2�е�װ�ý����ᴿ����ȴˮӦ��a�ڣ���a��b�����룮
��6����ʵ����ʹ�õ���ˮ�Ҵ��������46mL�������õ��Ĵ�������������50rnL����������IJ���Ϊ80.275%��
��Ҫ�Լ�������������
| ���� | ������ | ��״ | �ܶ�g/cm3 | �۵�� | �е�� | �ܽ�ȣ���/100ml�ܼ� | ||
| ˮ | �� | �� | ||||||
| ����ȩ | 106 | ��ɫҺ�� | 1.06 | -26 | 178-179 | 0.3 | ���� | ���� |
| ������ | 102 | ��ɫҺ�� | 1.082 | -73 | 138-140 | 12 | �� | ���� |
| ����� | 148 | ��ɫ�ᾧ | 1.248 | 133-134 | 300 | 0.04 | 24 | �� |
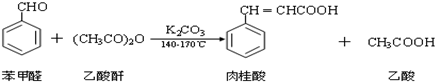
�ϳɣ���ͼ1��������������5.3g����ȩ��10.2g��������7.02g��ˮ̼��أ���140��170�棬���˻�������45min��
�������ᴿ���ٽ������ϳɵIJ�Ʒ��ȴ��߽������40mlˮ����5���ӣ�����ˮ�������ӻ�����г�ȥδ��Ӧ�ı���ȩ���õ��ֲ�Ʒ���ڽ������ֲ�Ʒ��ȴ�����40ml 10%����������ˮ��Һ���ټ�90mlˮ�����Ȼ���̿��ɫ�����ȹ��ˡ���ȴ���۽�1��1�������ڽ����¼��뵽���������Һ�У�����Һ�����ԣ�����ȴ����ѹ���ˡ�ϴ�ӡ�����Ȳ����õ��ϴ���������
ˮ��������ʹ�л�����ڽϵ͵��¶��´ӻ������������������Ա����ڳ�ѹ������ʱ����ɵ���ʧ����߷����ᴿ��Ч�ʣ�ͬʱ�ڲ�����װ�÷���Ҳ�ϼ�ѹ������һЩ������ˮ�����������Ӧ���ڷ�����ᴿ�л���ش��������⣺
��1���ϳ�������ʵ��������ˮ�����½��У�ʵ��ǰ������������ԭ������������ˮ�⣮ʵ����Ϊ�����¶���140��170������BD�м��ȣ�
A��ˮ B�����ͣ��е�290�棩 C��ɰ�� D��ֲ���ͣ��е�230��325�棩
��2��ͼ2��װ��a�������Dz���ˮ������װ��b�г���������Ҫ���������е�ԭ����ʹ�������ˮ������ֽӴ����������ᴿ����ᣬˮ����������Ʒ��������ƿ�����������ƣ���
��3�����ȹ��˵�Ŀ�ķ�ֹ������ƽᾧ����������©����
��4������1��1�������Ŀ����ʹ�������ת��Ϊ����ᣬ��������ᾧ����˺�ϴ�ӣ�ϴ�Ӽ���ˮ��
��5��5.3g����ȩ��10.2��������7.02g��ˮ̼��س�ַ�Ӧ�õ������ʵ��3.1g���������41.9%������3λ��Ч���֣���
��Ӧ�����вⶨ�IJ������ݼ��±�������t2��t1����
| ��Ӧʱ��/min | n��CO��/mol | n��H2O��/mol |
| 0 | 1.20 | 0.60 |
| t1 | 0.80 | |
| t2 | 0.20 |
| A�� | ��Ӧ��t1 min�ڵ�ƽ������Ϊv��H2��=$\frac{0.40}{{t}_{1}}$ mol•L-1•min-1 | |
| B�� | ���������������䣬��ʼʱ�������г���0.60 mol CO��1.20 mol H2O���ﵽƽ��ʱn��CO2��=0.40 mol | |
| C�� | ���������������䣬��ƽ����ϵ����ͨ��0.20 mol H2O����ԭƽ����ȣ��ﵽ��ƽ��ʱCOת���ʲ��䣬H2O������������� | |
| D�� | �¶�������800�棬������Ӧƽ�ⳣ��Ϊ0.64��������ӦΪ���ȷ�Ӧ |
 ���ܱ������У�ij��Ӧ�ڲ�ͬ��Ӧʱ������ʵ����ı仯�����ͼ��ʾ
���ܱ������У�ij��Ӧ�ڲ�ͬ��Ӧʱ������ʵ����ı仯�����ͼ��ʾ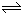 2C��
2C�� ��C�Ļ�ѧʽΪMg��OH��2
��C�Ļ�ѧʽΪMg��OH��2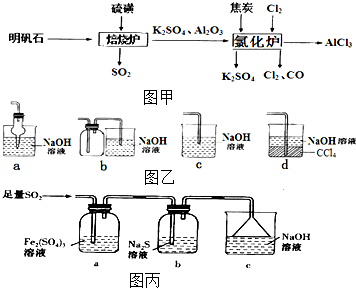 ��ˮ�Ȼ�����һ����Ҫ�Ļ���ԭ�ϣ���������ʯ[K2SO4•A12��SO4��3•2A12O3•6H2O]�Ʊ���ˮ�Ȼ�����������ͼ�ף�
��ˮ�Ȼ�����һ����Ҫ�Ļ���ԭ�ϣ���������ʯ[K2SO4•A12��SO4��3•2A12O3•6H2O]�Ʊ���ˮ�Ȼ�����������ͼ�ף�