��Ŀ����
ij����С�����������ʵ�鷽����֤Ag��ŨHNO3��Ӧ�Ĺ����п��ܲ��� NO����ʵ������ͼ���£�
NO����ʵ������ͼ���£�

�Ųⶨ��������ʵ���
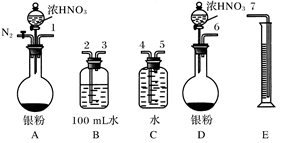
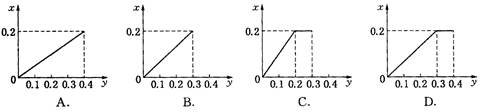
��Ӧ��������ͼBװ��������100 mL��Һ��ȡ��25.00 mL��Һ����0.1 mol��L��1��NaOH��Һ�ζ����÷�̪��ָʾ�����ζ�ǰ��ĵζ�����Һ���λ������ͼ��ʾ����B������������������ʵ���Ϊ____________����Ag��Ũ���ᷴӦ���������ɵ�NO2�����ʵ���Ϊ______________��
�ƲⶨNO�����
�ٴ���ͼ��ʾ��װ���У�����ΪӦѡ��________װ�ý���Ag��Ũ���ᷴӦʵ�飬ѡ�õ�������____________________________________________________________
________________________________________________________________________��
��ѡ����ͼ��ʾ�������һ�������ⶨ����NO�����װ�ã������������˳����_________________________(������ܿڱ��)��
���ڲⶨNO�����ʱ������Ͳ��ˮ��Һ��ȼ���ƿ��Һ��Ҫ�ͣ���ʱӦ����Ͳ��λ��______(ѡ��½��������ߡ�)���Ա�֤��Ͳ�е�Һ���뼯��ƿ�е�Һ���ƽ��
������ɷַ���
��ʵ����NO�����Ϊ112.0 mL(�����㵽��״��)����Ag��Ũ���ᷴӦ�Ĺ�����________(��С���û�С�)NO�����������жϵ�������__________________
________________________________________________________________________��
 NO����ʵ������ͼ���£�
NO����ʵ������ͼ���£�
�Ųⶨ��������ʵ���
��Ӧ��������ͼBװ��������100 mL��Һ��ȡ��25.00 mL��Һ����0.1 mol��L��1��NaOH��Һ�ζ����÷�̪��ָʾ�����ζ�ǰ��ĵζ�����Һ���λ������ͼ��ʾ����B������������������ʵ���Ϊ____________����Ag��Ũ���ᷴӦ���������ɵ�NO2�����ʵ���Ϊ______________��

�ƲⶨNO�����
�ٴ���ͼ��ʾ��װ���У�����ΪӦѡ��________װ�ý���Ag��Ũ���ᷴӦʵ�飬ѡ�õ�������____________________________________________________________
________________________________________________________________________��
��ѡ����ͼ��ʾ�������һ�������ⶨ����NO�����װ�ã������������˳����_________________________(������ܿڱ��)��
���ڲⶨNO�����ʱ������Ͳ��ˮ��Һ��ȼ���ƿ��Һ��Ҫ�ͣ���ʱӦ����Ͳ��λ��______(ѡ��½��������ߡ�)���Ա�֤��Ͳ�е�Һ���뼯��ƿ�е�Һ���ƽ��
������ɷַ���
��ʵ����NO�����Ϊ112.0 mL(�����㵽��״��)����Ag��Ũ���ᷴӦ�Ĺ�����________(��С���û�С�)NO�����������жϵ�������__________________
________________________________________________________________________��
(14��)��0.008 mol ��2�֣� 0.012 mol ��1�֣�
Bװ���з����ķ�Ӧ��3NO2��H2O===2HNO3��NO����NaOH��Һ�ζ�����HNO3����n(HNO3)��4n(NaOH)��4��0.1 mol��L��1��(20.40��0.40)��10��3 L��0.008 mol��������NO2�����ʵ���Ϊ��0.008 mol����0.012 mol��
�Ƣ�A��2�֣�
��ΪAװ�ÿ���ͨ��N2��װ���еĿ����ž�����ֹNO��������O2������2�֣�
��123547��2�֣���
������ ��1�֣�
���У�2�֣�
��ΪNO2��ˮ��Ӧ���ɵ�NO�����С���ռ�����NO�����(89.6<112.0)��2�֣�
Bװ���з����ķ�Ӧ��3NO2��H2O===2HNO3��NO����NaOH��Һ�ζ�����HNO3����n(HNO3)��4n(NaOH)��4��0.1 mol��L��1��(20.40��0.40)��10��3 L��0.008 mol��������NO2�����ʵ���Ϊ��0.008 mol����0.012 mol��
�Ƣ�A��2�֣�
��ΪAװ�ÿ���ͨ��N2��װ���еĿ����ž�����ֹNO��������O2������2�֣�
��123547��2�֣���
������ ��1�֣�
���У�2�֣�
��ΪNO2��ˮ��Ӧ���ɵ�NO�����С���ռ�����NO�����(89.6<112.0)��2�֣�
��

��ϰ��ϵ�д�
�����Ŀ

 �������м����һ�������̱����ô����������й�ʵ��ʱ������װ�ü��������㡢�������ԡ��ɷ���ʹ�õ��ŵ㡣�ô��������ܷ������е�ʵ����
�������м����һ�������̱����ô����������й�ʵ��ʱ������װ�ü��������㡢�������ԡ��ɷ���ʹ�õ��ŵ㡣�ô��������ܷ������е�ʵ����