��Ŀ����
����12�֣��ۻ�(As4S4)�ʹƻ�(As2S3)����ȡ�����Ҫ����ԭ�ϣ���������Ȼ���й����������������������գ�
��2��������Ӧ�еĻ�ԭ���� ��Ϊ�˷�ֹ������Ⱦ����Ӧ������������� ���ա�
��3��As2S3��HNO3�����·�Ӧ��As2S3+ 10H++ 10 NO��3 = 2H3AsO4+ 3S��+10NO2��+ 2H2O
������1mol NO2����Ӧ��ת�Ƶĵ�����Ϊ �������÷�Ӧ��Ƴ�һԭ��أ���NO2Ӧ���� ����������������������ݳ���
��4������Ӧ����NO2��11.2L O2����״������Ϻ���ˮ����ȫ��ת����ŨHNO3��Ȼ���������̼��Ӧ����������CO2���� ��ѡ���ţ���
a������0.5 mol b��С��0.5 mol c������0.5mol d����ȷ��
��2��������Ӧ�еĻ�ԭ���� ��Ϊ�˷�ֹ������Ⱦ����Ӧ������������� ���ա�
��3��As2S3��HNO3�����·�Ӧ��As2S3+ 10H++ 10 NO��3 = 2H3AsO4+ 3S��+10NO2��+ 2H2O
������1mol NO2����Ӧ��ת�Ƶĵ�����Ϊ �������÷�Ӧ��Ƴ�һԭ��أ���NO2Ӧ���� ����������������������ݳ���
��4������Ӧ����NO2��11.2L O2����״������Ϻ���ˮ����ȫ��ת����ŨHNO3��Ȼ���������̼��Ӧ����������CO2���� ��ѡ���ţ���
a������0.5 mol b��С��0.5 mol c������0.5mol d����ȷ��
����12�֣�ÿ��2�֣�
��1��2As2S3+ 2SnCl2 +4HCl= As4S4 +2 SnCl4 + 2H2S��
��2��SnCl2 ����������Һ������ͭ��Һ
��3��6.02��1022 ����
��4��b
��1��2As2S3+ 2SnCl2 +4HCl= As4S4 +2 SnCl4 + 2H2S��
��2��SnCl2 ����������Һ������ͭ��Һ
��3��6.02��1022 ����
��4��b
��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
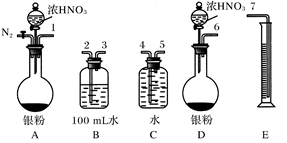
 NO����ʵ������ͼ���£�
NO����ʵ������ͼ���£�