ĚâÄżÄÚČÝ
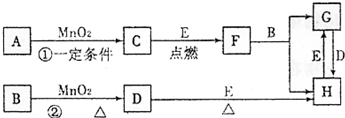
ĎÂÍĽËůÉ漰µÄÎďÖĘľůÎŞÖĐѧ»ŻŃ§ÖеijŁĽűÎďÖĘŁ¬ĆäÖĐ
Ł¨1Ł©Cˇ˘Dˇ˘Eˇ˘F¶ĽÎ޵ĄÖĘŁ¬ĆäÓŕÎŞ»ŻşĎÎď
Ł¨2Ł©Cˇ˘Dˇ˘E ÔÚłŁÎÂĎÂÎŞĆřĚ壬ĆäËüÎďÖĘÎŞąĚĚĺ»ňŇşĚĺ
Ł¨3Ł©Aˇ˘I Á˝ÖÖ»ŻşĎÎďµÄŃćÉ«·´Ó¦·Ö±đÎŞ×ĎÉ«şÍ»ĆÉ«
Ł¨4Ł©·´Ó¦ ˘Ů ˘Ú µÄһЩÉúłÉÎďŇŃľÂÔČĄŁ¬ÓĐĐ©·´Ó¦µÄĚőĽţδÁĐłö,ËüĂÇ´ćÔÚČçĎÂת»ŻąŘϵˇŁ
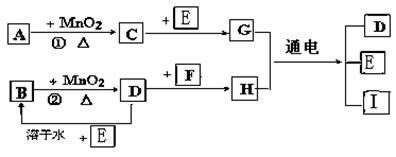 |
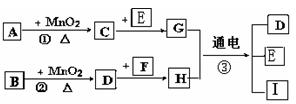
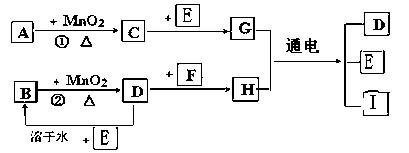
Ł¨1Ł©Đ´łöÓĐąŘÎďÖʵĻŻŃ§Ę˝ŁşAˇˇˇˇˇˇˇˇˇˇ Fˇˇˇˇˇˇˇˇˇˇ ˇŁ
Ł¨2Ł©Ö¸łöMnO2ÔÚĎŕąŘ·´Ó¦ÖеÄ×÷ÓĂŁş·´Ó¦ ˘Ů ÖĐĘǡˇˇˇˇˇ ĽÁŁ¬˘Ú ÖĐĘǡˇˇˇˇˇˇˇˇˇ ĽÁˇŁ
Ł¨3Ł©Đ´łö·´Ó¦˘Ú»ŻŃ§·˝łĚĘ˝ˇˇˇˇˇˇˇˇˇˇ ______ BÔÚ·´Ó¦ÖĐĎÔĘľˇˇˇˇˇˇ ĐԺ͡ˇˇˇˇˇ ĐÔ
Ł¨4Ł©Đ´łö·´Ó¦˘ŰµÄŔë×Ó·˝łĚĘ˝______________________________
Ł¨1Ł© KClO3ˇˇˇˇ ˇ˘ˇˇ Naˇˇ Ł¨2Ł©ˇˇ ´ß»ŻĽÁˇˇˇˇˇˇˇˇˇˇ ˇ˘ˇˇˇˇ Ńő»ŻĽÁˇˇˇˇˇˇˇˇˇˇˇˇ
Ł¨3Ł©ˇˇ MnO2+4HClŁ¨Ĺ¨Ł©MnCl2+Cl2+2H2Oˇˇˇˇ Ł» »ąÔĐÔˇˇˇˇ Ł» ËáĐÔˇˇˇˇˇˇˇˇˇˇˇˇˇˇˇˇˇˇ
Ł¨4Ł©ˇˇ 2Cl-+2H2O 2OH-
![]() +H2ˇü+Cl2ˇüˇˇˇˇ
+H2ˇü+Cl2ˇüˇˇˇˇ

Á·Ď°˛áϵÁĐ´đ°¸
 ĆÚÄ©łĺ´Ě100·Ö´´Đ½đľíÍęČ«ĘÔľíϵÁĐ´đ°¸
ĆÚÄ©łĺ´Ě100·Ö´´Đ½đľíÍęČ«ĘÔľíϵÁĐ´đ°¸
ĎŕąŘĚâÄż