��Ŀ����
��12�֣�ʳ��Ϊ��������ƴ����֡����ұ��涨����ʳ�����Ậ�����õ���3.5g/100mL��
��1��ij�о�С�����õζ��������ⶨijƷ��ʳ���д���ĺ���������˵����ȷ���� ��
a.��NaOH��Һ�ζ�ʱ��Ӧ�����ӷ���ʽΪ��H++OH-=H2O
b.ʳ��������ϡ��һ���������ٽ��еζ�
c.��NaOH��Һ�ζ�ʳ�ף���ʹ�÷�̪�������ָʾ��
d.����ø�Ʒ��ʳ�����ʵ���Ũ��Ϊ0.75mol��L-1�����ʳ��������Ϊ4.5g/100mL��
��2���о�С���ͬѧ��ϸ�۲��˸�Ʒ��ʳ�ı�ǩ���������л����б���������ΪʳƷ���Ӽ������������Ϸ���֤������ʳƷ���Ӽ�����������C6H5COONa�����ᷢ�����ӻ�����Ӧ���������һ���¶��µĴ����뱽����� ����ѡ����ĸ����
a.pH b.����� c.���볣�� d.�ܽ��
��3�������£���0.1000 mol��L-1NaOH��Һ�ֱ�ζ�20.00mL 0.1000 mol��L-1HCl��Һ��20.00mL 0.1000 mol��L-1CH3COOH��Һ���õ�2���ζ����ߣ�����ͼ��ʾ��
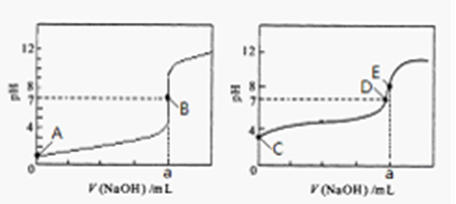
ͼ3 ͼ4
�ٵζ�������Һ�������� ���ͼl����ͼ2�������ζ�������a= mL
��4���������Բⶨһδ֪Ũ�ȵ�CH3COOH��Һ�ĵ���ȣ�Ӧ����ʵ���������Լ�����ֽ���� ��
a.��⣬NaOH��Һ b.����Na2CO3��Һ
c.������Ӧ��ʯ����Һ d.�к͵ζ���pH��ֽͼ2
(5)��ʽ�ζ���������ˮ��ϴ��δ�ñ�Һ��ϴ�����µζ����(�ƫС������ƫ����Ӱ�족) ����ʽ�ζ��ܶ���ʱ�����ȸ������������� ��
��1��ij�о�С�����õζ��������ⶨijƷ��ʳ���д���ĺ���������˵����ȷ���� ��
a.��NaOH��Һ�ζ�ʱ��Ӧ�����ӷ���ʽΪ��H++OH-=H2O
b.ʳ��������ϡ��һ���������ٽ��еζ�
c.��NaOH��Һ�ζ�ʳ�ף���ʹ�÷�̪�������ָʾ��
d.����ø�Ʒ��ʳ�����ʵ���Ũ��Ϊ0.75mol��L-1�����ʳ��������Ϊ4.5g/100mL��
��2���о�С���ͬѧ��ϸ�۲��˸�Ʒ��ʳ�ı�ǩ���������л����б���������ΪʳƷ���Ӽ������������Ϸ���֤������ʳƷ���Ӽ�����������C6H5COONa�����ᷢ�����ӻ�����Ӧ���������һ���¶��µĴ����뱽����� ����ѡ����ĸ����
a.pH b.����� c.���볣�� d.�ܽ��
��3�������£���0.1000 mol��L-1NaOH��Һ�ֱ�ζ�20.00mL 0.1000 mol��L-1HCl��Һ��20.00mL 0.1000 mol��L-1CH3COOH��Һ���õ�2���ζ����ߣ�����ͼ��ʾ��

ͼ3 ͼ4
�ٵζ�������Һ�������� ���ͼl����ͼ2�������ζ�������a= mL
��4���������Բⶨһδ֪Ũ�ȵ�CH3COOH��Һ�ĵ���ȣ�Ӧ����ʵ���������Լ�����ֽ���� ��
a.��⣬NaOH��Һ b.����Na2CO3��Һ
c.������Ӧ��ʯ����Һ d.�к͵ζ���pH��ֽͼ2
(5)��ʽ�ζ���������ˮ��ϴ��δ�ñ�Һ��ϴ�����µζ����(�ƫС������ƫ����Ӱ�족) ����ʽ�ζ��ܶ���ʱ�����ȸ������������� ��
��1��bd ��2��c ��3����ͼ2 20.00mL ��4��d (5)ƫ�� ƫ��
��1�����������ᣬ���ӷ���ʽ���û�ѧʽ��ʾ��a����ȷ��Ũ�ȹ���Ӧ���죬�������жϵζ��յ㣬���Կ���ͨ��ϡ�������ͷ�Ӧ���ʣ�b��ȷ��������������Ʒ�Ӧ���յ�ʱ��Һ�Լ��ԣ����ѡ���̪��ָʾ�������ü��ȡ����ʵ���Ũ��Ϊ0.75mol��L-1�����ʳ��������Ϊ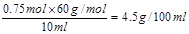 ��d��ȷ��
��d��ȷ��
��2�����ݽ�ǿ����ȡ������ԭ�����жϣ���ͨ���Ƚ϶��ߵĵ��볣�����Ƚ�������ǿ����
��3���������������ǡ�÷�Ӧʱ��Һ�����ԣ����������������ǡ�÷�Ӧʱ��Һ�Լ��ԣ�����ͼ2��ʾ����ĵζ����ߡ�
��4���������ָ��һ���¶��£��ѵ���ĵ���ʷ�����ռԭ���ܷ������������ѵ���ĺ�δ����ģ��İٷ��������Ҫ�ⶨ����ĵ���ȣ���Ҫ�ⶨ��������ʵ���Ũ���Լ��ѵ���Ĵ�������ʵ���Ũ�ȡ����ݵ��뷽��ʽ��֪����ͨ���ⶨ��Һ��pHֵ���ⶨ�ѵ���Ĵ�������ʵ���Ũ�ȡ�
��5����ʽ�ζ���������ˮ��ϴ��δ�ñ�Һ��ϴ�����Һ��ϡ�ͣ����ƫ�����ȸ������������ĵ����ƫ�����ƫ��
 ��d��ȷ��
��d��ȷ����2�����ݽ�ǿ����ȡ������ԭ�����жϣ���ͨ���Ƚ϶��ߵĵ��볣�����Ƚ�������ǿ����
��3���������������ǡ�÷�Ӧʱ��Һ�����ԣ����������������ǡ�÷�Ӧʱ��Һ�Լ��ԣ�����ͼ2��ʾ����ĵζ����ߡ�
��4���������ָ��һ���¶��£��ѵ���ĵ���ʷ�����ռԭ���ܷ������������ѵ���ĺ�δ����ģ��İٷ��������Ҫ�ⶨ����ĵ���ȣ���Ҫ�ⶨ��������ʵ���Ũ���Լ��ѵ���Ĵ�������ʵ���Ũ�ȡ����ݵ��뷽��ʽ��֪����ͨ���ⶨ��Һ��pHֵ���ⶨ�ѵ���Ĵ�������ʵ���Ũ�ȡ�
��5����ʽ�ζ���������ˮ��ϴ��δ�ñ�Һ��ϴ�����Һ��ϡ�ͣ����ƫ�����ȸ������������ĵ����ƫ�����ƫ��

��ϰ��ϵ�д�
 ��ʦ�㾦�ִʾ��ƪϵ�д�
��ʦ�㾦�ִʾ��ƪϵ�д�
�����Ŀ
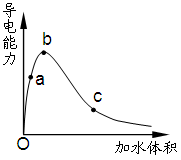


 H++HS-��HS-
H++HS-��HS-