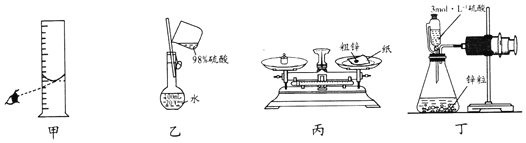
题目内容
【题目】下列实验操作能达到实验目的是( )
实验目的 | 实验操作 | |
A | 除去 | 向含有少量 |
B | 检验溶液中是否含有 | 取少量溶液于试管中,先加入 |
C | 比较HCl和 | 用pH试纸测定浓度均为 |
D | 验证 | 将硫酸酸化的 |
A.AB.BC.CD.D
【答案】A
【解析】
![]() 促进铁离子水解转化为沉淀,则反应后过滤可分离,故A正确;
促进铁离子水解转化为沉淀,则反应后过滤可分离,故A正确;
B.先加入![]() 溶液,可生成AgCl沉淀或硫酸钡沉淀,不能确定是否含硫酸根离子,故B错误;
溶液,可生成AgCl沉淀或硫酸钡沉淀,不能确定是否含硫酸根离子,故B错误;
C.NaClO溶液具有漂白性,不能选pH试纸测定pH,故C错误;
D.酸性条件下亚铁离子、硝酸根离子发生氧化还原反应,不能说明![]() 的氧化性比
的氧化性比![]() 强,故D错误;
强,故D错误;
答案选A。

【题目】甲醇是重要的化工原料。在催化剂的作用下,利用合成气(主要成分为CO、CO2和H2)合成甲醇的主要化学反应如下:
Ⅰ.CO+2H2![]() CH3OH
CH3OH
Ⅱ.CO2+3H2![]() CH3OH+H2O
CH3OH+H2O
Ⅲ.CO2+H2![]() CO+H2O
CO+H2O
请回答下列问题:
(1)已知三种物质的标准燃烧热如下表:
物质 | CO(g) | H2(g) | CH3OH(l) |
燃烧热(kJ·mol1) | 283.0 | 285.8 | 726.5 |
①书写25℃、101kPa条件时CO燃烧热的热化学方程式____________。
②计算25℃、101kPa条件时反应Ⅰ的ΔH=_____kJ·mol1 。
(2)甲醇燃料电池(Direct Methanol Fuel Cell)属于质子交换膜燃料电池,其工作原理如图所示:
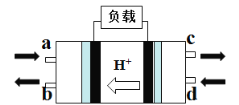
①c处产生的气体是_______________;
②负极的电极反应式是_______________。
(3)下图是甲醇燃料电池工作示意图,其中A,B,D均为石墨电极,C为铜电极。工作一段时间后,断开K,此时A,B两极上产生的气体体积相同。
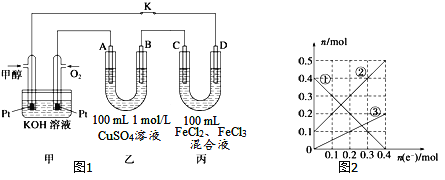
①乙中B极的电极反应式_______。
②乙中A极析出的气体在标况下的体积________。
③丙装置溶液中金属阳离子的物质的量与转移电子的物质的量变化关系如图,则②线表示的是____的变化,反应结束后,要使丙装置中金属阳离子恰好完全沉淀,需要____mL 5mol/LNaOH溶液。
(4)蓄电池是一种反复充电、放电的装置。有一种蓄电池在充电和放电时发生的反应如下:NiO2+Fe+2H2O![]() Fe(OH)2+Ni(OH)2。
Fe(OH)2+Ni(OH)2。
①此蓄电池在充电时,电池负极应与外加电源的________极连接。
②放电时正极的电极反应式为_____________________。
(5)按如图所示装置进行下列不同的操作

①将K1 、K2、 K3分别只关闭一个,则铁的腐蚀的速度最快的是只闭合_____(填“K1”或 “K2”或“K3”,下同),为减缓铁的腐蚀, 只闭合_____,该防护法称为______________。
②只闭合K3,当铁棒质量减少5.6g时,石墨电极消耗的气体体积为__________L(标准状况)。
【题目】根据下列操作及现象,所得结论不正确的是![]()
![]()
序号 | 操作及现象 | 结论 |
A | 将 |
|
B | “ |
|
C | 向25mL冷水和沸水中分别滴入5滴 | 温度升高, |
D | 将固体 | 同温下溶解度:
|
A. A B. B C. C D. D
【题目】Ⅰ.高铁酸钾(K2FeO4)具有极强的氧化性,是一种优良的水处理剂。
(1)请完成FeO42-与水反应的离子方程式:4FeO42-+10H2O![]() 4Fe(OH)3+8OH-+__。K2FeO4在处理水的过程中所起的作用是___和____。
4Fe(OH)3+8OH-+__。K2FeO4在处理水的过程中所起的作用是___和____。
(2)将适量K2FeO4配制成c(FeO42-)=1.0mmol·L-1的试样,将试样分别置于20℃、30℃、40℃和60℃的恒温水浴中,测定c(FeO42-)的变化,结果见图Ⅰ。第(1)题中的反应为FeO42-变化的主反应,则温度对该反应的反应速率和平衡移动的影响是___,发生反应的ΔH___0。

Ⅱ.下表是几种弱电解质的电离平衡常数、难溶电解质的溶度积Ksp(25℃)。
电解质 | 平衡方程式 | 平衡常数K | Ksp |
CH3COOH | CH3COOH | 1.76×10-5 | |
H2CO3 | H2CO3 HCO3- | K1=4.31×10-4 K2=5.61×10-11 | |
C6H5OH | C6H5OH | 1.1×10-10 | |
H3PO4 | H3PO4 H2PO4- | K1=7.52×10-3 K2=6.23×10-8 K3=2.20×10-13 | |
NH3·H2O | NH3·H2O | 1.76×10-5 | |
BaSO4 | BaSO4 | 1.07×10-10 | |
BaCO3 | BaCO3 | 2.58×10-9 |
回答下列问题:
(1)由上表分析,若①CH3COOH,②HCO3-,③C6H5OH,④H2PO4-均可看做酸,则它们酸性由强到弱的顺序为___(填编号)。
(2)写出C6H5OH与Na3PO4反应的离子方程式___。
(3)25℃时,将等体积等浓度的醋酸和氨水混合,混合液中:c(CH3COO-)__c(NH4+)(填“>”、“=”或“<”)。
(4)25℃时,向10mL0.01mol·L-1苯酚溶液中滴加VmL0.01mol·L-1氨水,混合液中粒子浓度关系正确的是__。
A.若混合液pH>7,则V≥10
B.若混合液pH<7,则c(NH4+)>c(C6H5O-)>c(H+)>c(OH-)
C.V=10时,混合液中水的电离程度小于10mL0.01mol·L-1苯酚溶液中水的电离程度
D.V=5时,2c(NH3·H2O)+2c(NH4
(5)水解反应是典型的可逆反应,水解反应的化学平衡常数称为水解常数(用Kb表示),类比化学平衡常数的定义,请写出Na2CO3第一步水解反应的水解常数的表达式:___。