��Ŀ����
����Ŀ��Ŀǰȫ��Χ�ڻ�����Ⱦ���ܹ�ע������������Ӧ������������ɫ��ѧ������ԭ�Ӿ������������ҵ�����еķ���������Ϊ��Ҫ����ҵ������������ʱ����ͬʱ������һ�ֳ�������Ҫ������������Ư����������(NaClO2)���乤���������£�
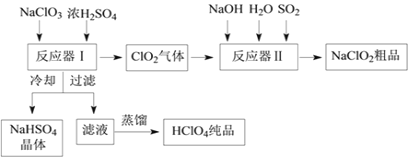
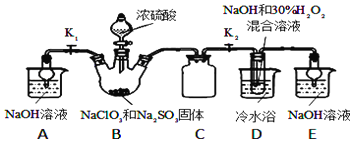
��֪����NaHSO4�ܽ�����¶ȵ����߶������ʵ������¿ɽᾧ�������ڸ�����������Ϊֹ������֪���е���ǿ�ᣬ�е�90������ش��������⣺
��1����Ӧ�����з�����Ӧ�Ļ�ѧ����ʽΪ_______����ȴ��Ŀ����____________��
��2����Ӧ�����з�����Ӧ�����ӷ���ʽΪ__________________________��
��3��ͨ�뷴Ӧ�����е�SO2��H2O2����ͬ��������NaClO2�����Ҫ˵��˫��ˮ�ڷ�Ӧ���ܴ���SO2��ԭ����_________________________��
��4��Ca(ClO)2��ClO2��NaClO2�Ⱥ��Ȼ����ﶼ�dz��õ���������Ư������д����ҵ������������ʯ������Ư�۾��Ļ�ѧ����ʽ��___________________________________��
���𰸡� 3NaClO3+3H2SO4��HClO4+2ClO2��+3NaHSO4+H2O ����NaHSO4���ܽ�ȣ�ʹNaHSO4�ᾧ���� 2ClO2+SO2+4OH����2ClO2��+SO42��+2H2O H2O2�л�ԭ��Ҳ�ܰ�ClO2��ԭΪNaClO2 2Cl2+2 Ca(OH )2��CaCl2+ Ca(ClO)2+2H2O
����������1�����ݼ��뵽��Ӧ�����е�ԭ����NaClO3��Ũ���ᣬ��������ClO2���塢NaHSO4��HClO4��Һ��������д��ѧ����ʽ�Ĺ���������ѧ��Ӧʱ����ԭ�ӵ��������Ŀ���䣬��д����ѧ��Ӧ����ʽΪ��3NaClO3+3H2SO4=HClO4+2ClO2+3NaHSO4+H2O��Ȼ��Ӧ���������Һ��ȴ�õ�NaHSO4���壬˵���������Ƶ��ܽ�����¶ȵĽ��Ͷ���С��
��2������ͨ�뵽��Ӧ�������еķ�Ӧ����NaOH��SO2��H2O��ClO2����������NaClO2���ȵĻ��ϼ���+4�۱����+3�ۣ��õ����ӣ�����������ԭ��Ӧ��ԭ����Ӧʧ���ӣ���+4�۱��+6�ۣ�����������ԭ��Ӧԭ�����������õ������������ڻ�ԭ��ʧȥ���ӵ���������������ԭ��Ӧ����ʽΪ2ClO2+SO2+4OH-=2ClO2-+SO42-+2H2O��
��3���������Ӧ�����е�SO2��H2O2����ͬ��������NaClO2��˵���任���ʺ�������ԭ��Ӧ�����ܷ�������NaClO2��Ҳ��˵��H2O2Ҳ���л�ԭ�ԣ�Ҳ�ܰ�ClO2��ԭΪNaClO2��
��4����������Ư��������ԭ����Ư��ԭ�����������ǵ�ǿ���������ﵽĿ�ģ����ݽ̲�֪ʶ��ҵ����ȡƯ��������������ʯ�ҷ�Ӧ�õ��ģ��䷴Ӧԭ��Ϊ2Cl2+2 Ca(OH )2��CaCl2+ Ca(ClO)2+2H2O��

 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�