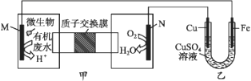
��Ŀ����
����Ŀ������ѧѡ��3:���ʽṹ�����ʡ�A��B��C��D��E����Ԫ�ص�ԭ�����������������зǽ���Ԫ��A�Ļ�̬ԭ���гɶԵ�������δ��ʱ��������������CԪ���ڵؿ��к�����ߣ�D�ĵ����Ƕ��������۵���͵Ľ�����E�ĺϽ����ҹ�ʹ������ĺϽ�
��1��EԪ�صĻ�̬ԭ�ӵ����Ų�ʽΪ__________________��
��2��A��ij���⻯��A2H2�����к���___���Ҽ���____���м���
��3��A �ĺ����������AO3n-�Ŀռ乹����___________��
��4��B������⻯��ķе��A ������⻯��ķе�ߵö࣬��ԭ����_____��
��5��E������������Ӧ��ˮ�����ܽ��ڰ�ˮ�����ɵĸ��ӻ�����Ļ�ѧʽ��______��
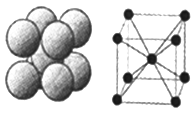
��6����ͼ��D���ʵľ���ѻ���ʽ�����ֶѻ���ʽ�ľ�����ԭ�ӵ���λ��Ϊ____������ԭ�ӵİ뾶Ϊrpm ���˾�����ܶȦ�=______g/cm3���ú�r�Ĵ���ʽ��ʾ������٤��������NA��ʾ����
���𰸡� 1s22s22p63s23p63d104s1 3 2 �������� �����Ӽ�����γ������������Ӽ䲻�ܣ������ķе�ȼ���� [Cu(NH3)4](OH)2 8 ![]()
�����������������A��B��C��D��E����Ԫ�ص�ԭ��������������CԪ���ڵؿ��к�����ߣ�C����Ԫ�ء����зǽ���Ԫ��A�Ļ�̬ԭ���гɶԵ�������δ��ʱ��������������ԭ������С����Ԫ�أ���A��̼Ԫ�أ�����B�ǵ�Ԫ�ء�D�ĵ����Ƕ��������۵���͵Ľ�������D���ơ�E�ĺϽ����ҹ�ʹ������ĺϽ���E��ͭ���ݴ˷������
������A��B��C��D��E����Ԫ�ص�ԭ��������������CԪ���ڵؿ��к�����ߣ�C����Ԫ�ء����зǽ���Ԫ��A�Ļ�̬ԭ���гɶԵ�������δ��ʱ��������������ԭ������С����Ԫ�أ���A��̼Ԫ�أ�����B�ǵ�Ԫ�ء�D�ĵ����Ƕ��������۵���͵Ľ�������D���ơ�E�ĺϽ����ҹ�ʹ������ĺϽ���E��ͭ��
��1��ͭ��ԭ��������29��ͭԪ�صĻ�̬ԭ�ӵ����Ų�ʽΪ1s22s22p63s23p63d104s1��
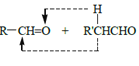
��2��A��ij���⻯��A2H2��������Ȳ����Ȳ�ĽṹʽΪH��C��C��H��������к���3��������2��������
��3��A �ĺ����������CO32-��̼ԭ�ӵļ۲���Ӷ�����![]() ������̼����Ŀռ乹����ƽ���������Ρ�
������̼����Ŀռ乹����ƽ���������Ρ�
��4�������Ӽ�����γ������������Ӽ䲻�ܣ������ķе�ȼ������
��5��ͭ������������Ӧ��ˮ����������ͭ�ܽ��ڰ�ˮ�����ɵĸ��ӻ�����Ļ�ѧʽ��[Cu(NH3)4](OH)2��
��6�����ݼصľ����ṹ��֪���ֶѻ���ʽ�ľ�����ԭ�ӵ���λ��Ϊ8������ԭ�ӵİ뾶Ϊrpm�������ĵĶԽ�����4rpm�����Ծ����ı߳���![]() ���þ����������
���þ����������![]() �������к���2����ԭ�ӣ���˾�����ܶ�����
�������к���2����ԭ�ӣ���˾�����ܶ�����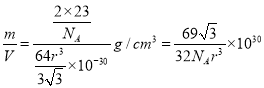 g/cm3��
g/cm3��

 ���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д�
���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д�