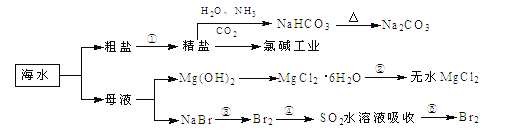
ΧβΡΩΡΎ»ί
ΓΨΧβΡΩΓΩΈΣ―–ΨΩΚΘΥ°ΧαδεΙΛ“’Θ§ΦΉΓΔ““ΝΫΆ§―ßΖ÷±π…ηΦΤΝΥ»γœ¬ Β―ιΝς≥ΧΘΚ
ΦΉΘΚ![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
““ΘΚ![]()
![]()
![]()
![]()
![]()
![]()
![]()
Θ®1Θ©ΦΉΓΔ““ΝΫΆ§―ß‘ΎΒΎ“ΜΫΉΕΈΒΟΒΫΚ§δεΚΘΥ°÷–Θ§¬»ΤχΒΡάϊ”Ο¬ ΫœΗΏΒΡ «________(ΧνΓΑΦΉΓ±ΜρΓΑ““Γ±)Θ§‘≠“ρ «____________________________________ΓΘ
Θ®2Θ©ΦΉΆ§―ß≤Ϋ÷ηΔήΥυΖΔ…ζΖ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣ________________________ΓΘ
Θ®3Θ©Ε‘±»ΦΉΓΔ““ΝΫΝς≥ΧΘ§Ήν¥σΒΡ«χ±π‘Ύ”ΎΕ‘Κ§δεΚΘΥ°ΒΡ¥ΠάμΖΫΖ®≤ΜΆ§Θ§Τδ÷–ΖϊΚœΙΛ“Β…ζ≤ζ“Σ«σΒΡ «________(ΧνΓΑΦΉΓ±ΜρΓΑ““Γ±)Θ§άμ”… «__________________________________ΓΘ
Θ®4Θ©Ρ≥ΩΈΆβ–ΓΉι‘Ύ Β―ι “ΡΘΡβΙΛ“Β…œ¥”≈®ΥθΚΘΥ°÷–Χα»ΓδεΒΡΙΛ“’Νς≥ΧΘ§…ηΦΤ“‘œ¬ΉΑ÷ΟΫχ–– Β―ιΘ®Υυ”–œπΫΚ÷ΤΤΖΨυ“―±Μ±ΘΜΛΘ§Φ–≥÷ΉΑ÷Ο“―¬‘»ΞΘ©ΓΘœ¬Ν–ΥΒΖ®¥μΈσΒΡ «___________ΓΘ
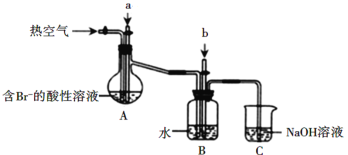
AΘ°AΉΑ÷Ο÷–Ά®»κΒΡaΤχΧε «Cl2
BΘ° Β―ι ±”Π‘ΎAΉΑ÷Ο÷–Ά®»κaΤχΧε“ΜΕΈ ±ΦδΚσΘ§ΆΘ÷ΙΆ®»κΘ§ΗΡΆ®»κ»»Ω’Τχ
CΘ°BΉΑ÷Ο÷–Ά®»κΒΡbΤχΧε «SO2
DΘ°CΉΑ÷ΟΒΡΉς”Ο÷Μ «Έϋ ’Εύ”ύΒΡSO2ΤχΧε
ΓΨ¥πΑΗΓΩΦΉ ΥαΜ·Ω…Ζά÷ΙCl2”κH2OΖ¥”Π SO2ΘΪBr2ΘΪ2H2O===4H+ΘΪSO42-ΘΪ2BrΘ≠ ΦΉ Κ§δεΚΘΥ°÷–δεΒΡ≈®Ε»ΒΆΘ§÷±Ϋ”’τΝσ≥…±ΨΗΏΘ§ΦΉΝς≥Χ÷–ΔέΔήΔί≤Ϋ ΒΦ …œ «δεΒΡΗΜΦ·Ιΐ≥ΧΘ§Ω…ΧαΗΏδεΒΡ≈®Ε»Θ§Φθ…ΌΡή‘¥œϊΚΡΘ§ΫΒΒΆ≥…±Ψ D
ΓΨΫβΈωΓΩ
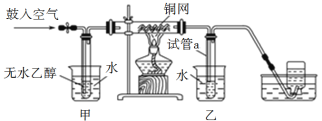
Θ®1Θ©¬»ΤχΆ®»κΒΫΚΘΥ°÷–Θ§ΖΔ…ζΖ¥”Π…ζ≥…―ΈΥαΓΔ¥Έ¬»ΥαΘ§»τ”ΟΝρΥαΥαΜ·Θ§‘ρ‘ω¥σ»ή“Κ÷–«βάκΉ”≈®Ε»Θ§ ΙΤΫΚβΡφœρ“ΤΕ·Θ§¥”ΕχΧαΗΏ¬»ΤχΒΡάϊ”Ο¬ ΘΜ
Θ®2Θ©≤Ϋ÷ηΔή”ΟΕΰ―θΜ·Νρ”κδεΖ¥”Π…ζ≥…ΝρΥαΚΆ«βδεΥαά¥Έϋ ’δε’τΤχΘ§Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣΘΚSO2ΘΪBr2ΘΪ2H2O=4H+ΘΪSO42-ΘΪ2BrΘ≠ΘΜ
Θ®3Θ©ΙΛ“ΒΝς≥Χ“Σ«σ Ήœ»Ε‘ ‘ΦΝΫχ––ΗΜΦ·Θ§¥σ≈ζΝΩ…ζ≤ζΘ§«“ΫΒΒΆ≥…±ΨΘ§‘ρΦΉΖϊΚœΘΜΙΛ“ΒΫχ––’τΝσ ±–η“Σ¥σΝΩΒΡΡή‘¥Θ§«“≤ζΝΩΒΆΘΜ
Θ®4Θ©AΘ°AΉΑ÷Ο÷–Ά®»κΒΡaΤχΧε «Cl2Θ§άϊ”Ο¬»ΤχΫΪ»ή“Κ÷–ΒΡδεάκΉ”―θΜ·ΈΣΒΞ÷ Θ§A’ΐ»ΖΘΜ
BΘ° Β―ι ±”Π‘ΎAΉΑ÷Ο÷–Ά®»κ¬»ΤχΤχΧε“ΜΕΈ ±ΦδΚσΘ§ΆΘ÷ΙΆ®»κΘ§ΗΡΆ®»κ»»Ω’ΤχΘ§Ζώ‘ρΆ®»κ»»Ω’ΤχΜαΫΒΒΆ¬»ΤχΒΡάϊ”Ο¬ Θ§«“≤ζ…欻Τχ‘”÷ Θ§B’ΐ»ΖΘΜ
CΘ°BΉΑ÷Ο÷–Ά®»κΒΡbΤχΧε «SO2Θ§”ΟΕΰ―θΜ·ΝρΈϋ ’Ω’Τχ÷–ΒΡδεΒΞ÷ Θ§Ϋχ––≤ζΤΖΒΡΗΜΦ·Θ§C’ΐ»ΖΘΜ
DΘ°CΉΑ÷ΟΒΡΉς”ΟΈϋ ’Εύ”ύΒΡSO2ΤχΧεΚΆΈ¥Ζ¥”ΠΒΡδε’τΤχΘ§D¥μΈσΘΜ
¥πΑΗΈΣD

ΓΨΧβΡΩΓΩΈΣ≥ΪΒΦΓΑΫΎΡήΦθ≈≈Γ±ΚΆΓΑΒΆΧΦΨ≠ΦΟΓ±Θ§ΡΩ«ΑΙΛ“Β…œ”–“Μ÷÷ΖΫΖ® «”ΟCO2ά¥…ζ≤ζ»ΦΝœ““¥ΦΓΘ“ΜΕ®ΧθΦΰœ¬ΖΔ…ζΖ¥”ΠΘΚ2CO2(g)ΘΪ6H2(g)![]() CH3CH2OH(g)ΘΪ3H2O(g)ΓΓΠΛH<0ΓΘ
CH3CH2OH(g)ΘΪ3H2O(g)ΓΓΠΛH<0ΓΘ
Θ®1Θ©‘Ύ“ΜΕ®ΧθΦΰœ¬Θ§‘Ύ20 LΟή±’»ίΤς÷–Α¥Έο÷ ΒΡΝΩ±»ΈΣ1ΓΟ3≥δ»κCO2ΚΆH2Θ§Έ¬Ε»‘Ύ450 KΘ§n(H2)Υφ ±Φδ±δΜ·»γ±μΥυ ΨΘΚ
t/min | 0 | 1 | 3 | 5 |
n(H2)/mol | 8 | 6 | 5 | 5 |
‘Ύ450 ΓφΓΔ0ΓΪ1 minΘ§v(CH3CH2OH)ΘΫ________ΘΜ¥ΥΈ¬Ε»œ¬ΗΟΖ¥”ΠΒΡΜ·―ßΤΫΚβ≥Θ ΐΈΣ________(ΫαΙϊ±ΘΝτ»ΐΈΜ”––ß ΐΉ÷)ΓΘ
Θ®2Θ©‘Ύ5 MPaœ¬≤βΒΟΤΫΚβΧεœΒ÷–ΗςΈο÷ ΒΡΧεΜΐΖ÷ ΐΥφΈ¬Ε»ΒΡ±δΜ·«ζœΏ»γΆΦΥυ ΨΘΚ
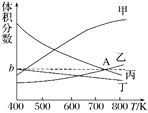
«ζœΏ““±μ ΨΒΡ « ________(ΧνΈο÷ ΒΡΜ·―ß Ϋ)ΒΡΧεΜΐΖ÷ ΐΘ§ΆΦœώ÷–AΒψΕ‘”ΠΒΡΧεΜΐΖ÷ ΐbΘΫ________%(ΫαΙϊ±ΘΝτ»ΐΈΜ”––ß ΐΉ÷)ΓΘ
Θ®3Θ©œ¬Ν–¥κ ©÷–Ρή ΙΜ·―ßΤΫΚβœρ’ΐΖ¥”ΠΖΫœρ“ΤΕ·ΒΡ «________ΓΘ
AΘ°…ΐΗΏΈ¬Ε»
BΘ°ΫΪCH3CH2OH(g)ΦΑ ±“ΚΜ·≥ι≥ω
CΘ°―Γ‘ώΗΏ–ߥΏΜ·ΦΝ
DΘ°‘Ό≥δ»κl mol CO2ΚΆ3 mol H2
Θ®4Θ©25 ΓφΓΔ1.01ΓΝ105Pa ±Θ§9.2 g“ΚΧ§““¥ΦΆξ»Ϊ»Φ…’Θ§Β±Μ÷Η¥ΒΫ‘≠Ή¥Χ§ ±Θ§Ζ≈≥ω273.4 kJΒΡ»»ΝΩΘ§–¥≥ω±μ Ψ““¥Φ»Φ…’ΒΡ»»Μ·―ßΖΫ≥Χ ΫΘΚ________________________ΓΘ
Θ®5Θ©“‘ ·ΡΪΈΣΒγΦΪΘ§«β―θΜ·ΡΤΓΔ““¥ΦΓΔΥ°ΓΔ―θΤχΈΣ‘≠ΝœΘ§Ω…“‘÷Τ≥…““¥ΦΒΡ»ΦΝœΒγ≥ΊΘ§–¥≥ωΖΔ…ζΜΙ‘≠Ζ¥”ΠΒΡΒγΦΪΖ¥”Π ΫΘΚ_____________________________________ΓΘ