��Ŀ����
13�������������ʻԱ�Ƿƺ��ʻ���������ٻ�ɫ�������ữ��CrO3��Һ�����Ҵ����������ɵ���ɫCr3+��Ũ����ȷ�����䷴Ӧԭ��ΪCrO3+CH3CH2OH+H2SO4��Cr2��SO4��3+CH3CHO+H2O��δ��ƽ��������˵����ȷ���ǣ�������| A�� | �÷�Ӧ��CH3CH2OH���������� | |
| B�� | �÷�Ӧ����������Ӧ�Ĺ�����CrO3��Cr2��SO4��3 | |
| C�� | �÷�Ӧ�����������뻹ԭ��������ʵ���֮��Ϊ2��3 | |
| D�� | �÷�Ӧ��ÿ����1mol H2Oת��1mol���� |
���� CrO3+CH3CH2OH+H2SO4��Cr2��SO4��3+CH3CHO+H2O�У�CrԪ�صĻ��ϼ���+6�۽���Ϊ+3�ۣ��Ҵ���CԪ�صĻ��ϼ���-2�۱�Ϊ-1�ۣ��÷�ӦΪ2CrO3+3C2H5OH+3H2SO4�TCr2��SO4��3+3CH3CHO+6H2O����CrԪ�صĻ��ϼ۱仯��֪ÿ����6mol H2Oת��6mol���ӣ��Դ������
��� �⣺A��CԪ�صĻ��ϼ����ߣ���CH3CH2OH���ֻ�ԭ�ԣ���A����
B��CrԪ�صĻ��ϼ۽��ͣ��õ����ӱ���ԭ����ԭ��ӦΪCrO3��Cr2��SO4��3����B����
C���ɷ�Ӧ2CrO3+3C2H5OH+3H2SO4�TCr2��SO4��3+3CH3CHO+6H2O��֪����������ΪCH3CHO����ԭ����Cr2��SO4��3�������������뻹ԭ��������ʵ���֮��Ϊ3��1����C����
D����CrԪ�صĻ��ϼ۱仯��֪ÿ����6mol H2Oת��6mol���ӣ���÷�Ӧ��ÿ����1mol H2Oת��1mol���ӣ���D��ȷ��
��ѡD��
���� ���⿼��������ԭ��Ӧ��Ϊ��Ƶ���㣬���շ�Ӧ��Ԫ�صĻ��ϼ۱仯Ϊ���Ĺؼ�������������ԭ��Ӧ�������ת�Ƶ��ӵĿ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д� �������ϵ�д�
�������ϵ�д�
�����Ŀ
3������ԭ�ӽṹ�������У�
������ԭ�Ӻ˾������Ӻ����ӹ��ɣ�
��ԭ�Ӵ��ڻ�̬ʱ���������Ӳ�һ����8����
��ϡ������Ԫ�أ����̬ԭ������㶼��8�����ӣ�
������������Ϊ2��ԭ��һ���ǽ���ԭ�ӣ�
������ȷ���ǣ�������
������ԭ�Ӻ˾������Ӻ����ӹ��ɣ�
��ԭ�Ӵ��ڻ�̬ʱ���������Ӳ�һ����8����
��ϡ������Ԫ�أ����̬ԭ������㶼��8�����ӣ�
������������Ϊ2��ԭ��һ���ǽ���ԭ�ӣ�
������ȷ���ǣ�������
| A�� | �٢� | B�� | �ڢ� | C�� | ֻ�Т� | D�� | ֻ�Т� |
4��ǿ������Һ���ܴ���������������ǣ�������
| A�� | Na+��Fe2+��Cl-��SO42- | B�� | K+��Ag+��Cl-��NO3- | ||
| C�� | Ba2+��Na+��NO3-��CO32- | D�� | Na+��K+��CO32-��SO32- |
8��ijѧ���������³�����Ʒ������NaHCO3��NaCl�����е��ǣ�������
| A�� | ʳ�� | B�� | ���� | C�� | ���� | D�� | �� |
5��ij��ɫ�Ļ��������ܺ���NO2��NO��CO2��O2��NH3��N2�еļ��֣���100mL�����徭����ͼ��ʾ��ʵ����д���������õ�������Һ��������������ʣ�࣬�йظ�������ɵ��жϲ���ȷ���ǣ�������
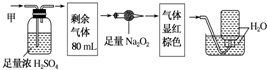

| A�� | һ����NH3�������Ϊ20mL | B�� | һ����CO2�������Ϊ48mL | ||
| C�� | һ����NO�������Ϊ30mL | D�� | ��������һ������NO2��N2��O2 |
2����һ������Cu��Fe2O3�Ļ�������м���300mL 1mol/L �����ᣬǡ��ʹ�������ȫ�ܽ⣬������Һ�в���Fe3+�����ù�����CO�ڸ����»�ԭ��ͬ������ԭ����������ٵ�����Ϊ��������
| A�� | 6.4 g | B�� | 4.8 g | C�� | 2.4 g | D�� | 1.6 g |
3�������������ʻ�Ϊͬ����������ǣ�������
��16O2��18O2 ��H2SiO3��H4SiO4 ��O2��O3 ��H2O��H2O2 �ݽ��ʯ��ʯī��C60 ���Ͱ��ף�
��16O2��18O2 ��H2SiO3��H4SiO4 ��O2��O3 ��H2O��H2O2 �ݽ��ʯ��ʯī��C60 ���Ͱ��ף�
| A�� | ֻ�Тڢۢ� | B�� | ֻ�Тۢݢ� | C�� | ֻ�Т٢ۢ� | D�� | ֻ�Т٢ڢܢ� |
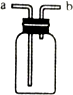 ��ͼװ������ѧ��ѧʵ���г�������������������ϴ���⣬����������;��
��ͼװ������ѧ��ѧʵ���г�������������������ϴ���⣬����������;��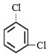
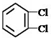 ����35Cl��37Cl��
����35Cl��37Cl��