��Ŀ����
��6�֣����ľ�������ظ�ԭ���۱��衱֮�����������⣬���и�������˾��ȵ��ο���Դ��Һ����Լؿ�Ϊ����������þ����ȶ��ֿ����ij�о���ѧϰС����ȡ�κ���±��Ũ��Һ������K+��Mg2+��Br-��SO42-��Cl-�ȣ�������ȡ�ϴ������Ȼ��ؾ��弰Һ�壨Br2��������������������̣�
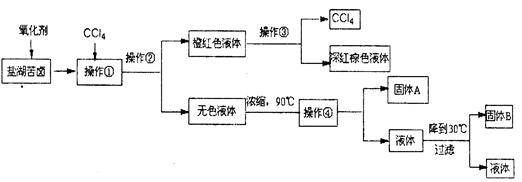
������������̣��ش�������⣺
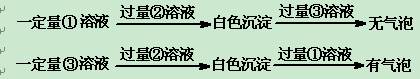
��1�������ڵ��������Ҫ������ ��
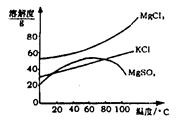
��2��������ͼ�ܽ�����ߣ��õ��Ĺ���A����Ҫ�ɷ��� ���ѧʽ����
��3��ͬѧ�����һЩ�µķ����������������ں���ɫ��Һ���г����ᴿ���䷽�����£�
���й����ϡ�
����Ƴ��ӹ��̡�
����֪�Լ�B��K2CO3��Һ������ҺA����Ҫ�ɷ��� ���ѧʽ����
����ȡ�����Ȼ��ء�
�ڶ���ҺB���Ȳ����ϵμ�l mol�� Lһ1��������Һ��ͬʱ��pH��ֽ�����Һ��ֱ��pH=5ʱֹͣ�����ᣬ�õ���ҺC���ò�����Ŀ���� ��
�۽���ҺC�����������У������������ò��������Ͻ��裬ֱ�� ʱ��������ֹͣ���ȡ�
���������ۡ�
�ܽ��в������п�����ҺpH=12��ȷ��Mg2+�����������ṩ�����ݼ��㣬��ʱ��ҺB��Mg2+���ʵ���Ũ��Ϊ ��

������������̣��ش�������⣺
��1�������ڵ��������Ҫ������ ��
��2��������ͼ�ܽ�����ߣ��õ��Ĺ���A����Ҫ�ɷ��� ���ѧʽ����

��3��ͬѧ�����һЩ�µķ����������������ں���ɫ��Һ���г����ᴿ���䷽�����£�
���й����ϡ�
| ��ѧʽ | BaCO3 | BaSO4 | Ca��OH��2 | MgCO3 | Mg��OH��2 |
| Ksp | 8.1��10һ9 | 1.08��10һ10 | 1.0��10һ4 | 3.5��10һ5 | 1.6��10һ11 |

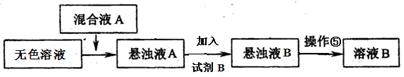
����֪�Լ�B��K2CO3��Һ������ҺA����Ҫ�ɷ��� ���ѧʽ����
����ȡ�����Ȼ��ء�
�ڶ���ҺB���Ȳ����ϵμ�l mol�� Lһ1��������Һ��ͬʱ��pH��ֽ�����Һ��ֱ��pH=5ʱֹͣ�����ᣬ�õ���ҺC���ò�����Ŀ���� ��
�۽���ҺC�����������У������������ò��������Ͻ��裬ֱ�� ʱ��������ֹͣ���ȡ�
���������ۡ�
�ܽ��в������п�����ҺpH=12��ȷ��Mg2+�����������ṩ�����ݼ��㣬��ʱ��ҺB��Mg2+���ʵ���Ũ��Ϊ ��
��1����Һ©�� ��2��MgSO4
��3����Ba(OH)2(��BaCl2��KOH)�ڳ�ȥδ��Ӧ��OH-��CO32-�۳��ֽ϶ྦྷ��,ʣ�����ˮʱ��1.6��10-7mol/L (ÿ��1��,��6��)
��3����Ba(OH)2(��BaCl2��KOH)�ڳ�ȥδ��Ӧ��OH-��CO32-�۳��ֽ϶ྦྷ��,ʣ�����ˮʱ��1.6��10-7mol/L (ÿ��1��,��6��)
��

��ϰ��ϵ�д�
 �����ѧСѧ�꼶�νӽݾ��㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӽݾ��㽭��ѧ������ϵ�д�
�����Ŀ

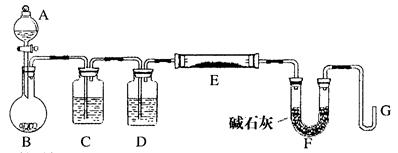


 ������
�����Ρ� ������д���пհף�
������д���пհף�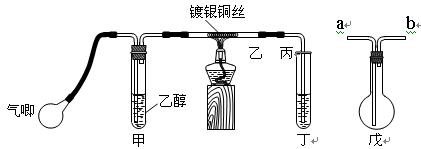
 ���� _____ ��
���� _____ ��
 ��
�� ��
�� �����ʣ�
�����ʣ� ��
�� [һֱ
[һֱ �����������������ܽ��ڰ�ˮ]��������д����ʵ�鱨�档
�����������������ܽ��ڰ�ˮ]��������д����ʵ�鱨�档
