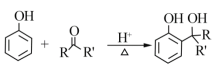
��Ŀ����
����Ŀ�������������ճ��������������Ź㷺��Ӧ�ã���ش��������⣺
��1����������ʹ�õĺϽ���___________��Ŀǰʹ�������Ľ�����________��
��2���������Ͻ����ճ������еij��ò��ϣ�����������ȷ����________��
A.������Ӳ�ȱ������� B.������ʴ��ǿ����������
C.����������Ͻ�ֻ������Ԫ�� D.����һ�������£�����ˮ������Ӧ
E.�������Ũ�����л�ۻ�
��3�����ˮ�е��뼸�α���FeCl3��Һ��������Һ������ĺ��ɫ���÷�Ӧ�����ӷ���ʽΪ__________���γɵķ�ɢϵ�з�ɢ�ʵ���ֱ����Χ��_________��
��4��������������Һ�м���������ƣ��к��ɫ�������ɣ���������Na2O2�����ɵ�O2�����ʵ���֮��Ϊ3��1����д�����������ӷ�Ӧ����ʽ___________________��
��5�����ӹ�ҵ��Ҫ��30%��FeCl3��Һ��ʴ��Ե���ϵ�ͭ������ӡˢ��·�塣��д��FeCl3��Һ��ͭ��Ӧ�����ӷ���ʽ��_________________����ʴ��ķ�Һ�м���һ���������۳�ַ�Ӧ������ʣ�࣬��Ӧ�����Һ��һ�����е�������___________�����ܺ��е�������_________������Ӹ�ʴ��ķ�Һ�л���ͭ�����»��FeCl3��Һ�����������Լ�����������������Ũ������Ũ�������ռ���Ũ��ˮ����Ҫ�õ���һ���Լ���_______��
A.�٢ڢ� B.�٢ۢܢ� C.�ڢܢ� D.�٢ܢ�
��6����������(Na2FeO4)��һ�����;�ˮ�����������Ƶ��Ʊ�����֮һ�ǣ��ڼ�����������NaClO����Fe3+����д���÷�Ӧ�����ӷ�Ӧ����ʽ______________��
��7����һ��������������Ʒ����5 mol/L����140 mL��ǡ����ȫ�ܽ⣬������Һ�������ձ����0.56 L������ǡ��ʹ����Fe2+ȫ��ת����Fe3+����������Ļ�ѧʽ��_____________��
���𰸡���1����ͭ ��
��2��BDE
��3��Fe3++3H2O![]() Fe(OH)3(����)+3H+ 1~100 nm
Fe(OH)3(����)+3H+ 1~100 nm
��4��3Na2O2+2Fe2++4H2O![]() 2Fe(OH)3��+O2��+6Na++2OH
2Fe(OH)3��+O2��+6Na++2OH
��5��2Fe3++Cu![]() 2Fe2++Cu2+ Fe2+��Cu2+��Cl Fe3+ A
2Fe2++Cu2+ Fe2+��Cu2+��Cl Fe3+ A
��6��2Fe(OH)3+3ClO+4OH![]() 2
2![]() +3Cl+5H2O
+3Cl+5H2O
��7��Fe5O7
�����������⿼�����Ԫ�ؼ��仯��������ʡ���1����������ʹ�õĺϽ�����ͭ��Ŀǰʹ�������Ľ���������
��2��A.������Ӳ�ȱ������ͣ�A����B.������ʴ��ǿ���������⣬B��ȷ��C.����������Ͻ𣬻����зǽ���Ԫ�أ�C����D.����һ�������£�����ˮ������Ӧ����������������������D��ȷ��E.�������������Ũ�����л�ۻ���E��ȷ����ѡBDE��
��3�����ˮ�е��뼸�α���FeCl3��Һ��������Һ������ĺ��ɫ�����������������壬�÷�Ӧ�����ӷ���ʽΪFe3++3H2O![]() Fe(OH)3(����)+3H+���γɵķ�ɢϵ�ǽ��壬���з�ɢ�ʵ���ֱ����Χ��1��100nm��
Fe(OH)3(����)+3H+���γɵķ�ɢϵ�ǽ��壬���з�ɢ�ʵ���ֱ����Χ��1��100nm��
��4��������������Һ�м���������ƣ��к��ɫ�������ɣ���������������������������Na2O2�����ɵ�O2�����ʵ���֮��Ϊ3��1�������ԭ���غ�͵��ӵ�ʧ�غ��֪���������ӷ�Ӧ����ʽΪ3Na2O2+2Fe2++4H2O![]() 2Fe(OH)3��+O2��+6Na++2OH��
2Fe(OH)3��+O2��+6Na++2OH��
��5��FeCl3��Һ��ͭ��Ӧ�����ӷ���ʽΪ2Fe3++Cu![]() 2Fe2++Cu2+����ʴ��ķ�Һ�м���һ���������۳�ַ�Ӧ������ʣ�࣬˵����ֻ�������ӷ�Ӧ����Ӧ�����Һ��һ�����е�������Fe2+��Cu2+��Cl�����ܺ��е�������Fe3+������Ӹ�ʴ��ķ�Һ�л���ͭ�����»��FeCl3��Һ������Ҫ������������û���ͭ�����˺������������ᣬȻ���ٹ��ˣ��ϲ���ͨ���������ɵõ��Ȼ�����Һ�������Ҫ���Լ�Ϊ�������������ۡ���Ũ���ᣬ��ѡA��
2Fe2++Cu2+����ʴ��ķ�Һ�м���һ���������۳�ַ�Ӧ������ʣ�࣬˵����ֻ�������ӷ�Ӧ����Ӧ�����Һ��һ�����е�������Fe2+��Cu2+��Cl�����ܺ��е�������Fe3+������Ӹ�ʴ��ķ�Һ�л���ͭ�����»��FeCl3��Һ������Ҫ������������û���ͭ�����˺������������ᣬȻ���ٹ��ˣ��ϲ���ͨ���������ɵõ��Ȼ�����Һ�������Ҫ���Լ�Ϊ�������������ۡ���Ũ���ᣬ��ѡA��
��6���ڼ�����������NaClO����Fe3+�������ɸ������ƣ��仹ԭ�����������ӣ�����ݵ��ӵ�ʧ�غ��ԭ���غ��֪���÷�Ӧ�����ӷ�Ӧ����ʽΪ2Fe(OH)3+3ClO+4OH![]() 2
2![]() +3Cl+5H2O��
+3Cl+5H2O��
��7����������ʵ�����0.7 mol������0.35 molˮ��������������ԭ�ӵ����ʵ�����0.35 mol��������0.025 mol������������Һ���Ȼ��������ʵ�����(0.70 mol+0.025 mol��2)��3=0.25 mol������ԭ�ӵ����ʵ�����0.25 mol��������ԭ�Ӻ���ԭ�ӵ����ʵ���֮����5�U7����˸�������Ļ�ѧʽ��Fe5O7��