��Ŀ����
����Ŀ����17�֣�
�Ǽ��㶹����һ�����Ƶ���ʯ��ҩ��ϳ�·������ͼ��ʾ��

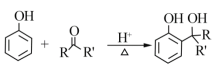
��֪��
RCOOR'+R'OH![]() RCOOR'+ R'OH��R��R'��R'����������
RCOOR'+ R'OH��R��R'��R'����������
��1��A���ڷ���������ṹ��ʽ��______________________��B�������Ĺ�������________________��
��2��C��D�ķ�Ӧ������___________________��
��3��E����֬�ࡣ�����Ҵ�Ϊ�л�ԭ�ϣ�ѡ�ñ�Ҫ�����Լ��ϳ�E��д���йػ�ѧ����ʽ��______________________________��
��4����֪��2E![]() F+C2H5OH��F������������
F+C2H5OH��F������������![]() ��___________��
��___________��
��5����D��FΪԭ�Ϻϳ��Ǽ��㶹�ط�Ϊ������Ӧ��д���йػ�����Ľṹ��ʽ��

���𰸡�
��1��![]() ��������
��������
��2��ȡ����
��3��2CH3CH2OH+O2 ![]() 2CH3CHO+2H2O 2CH3CHO+O2
2CH3CHO+2H2O 2CH3CHO+O2 ![]() 2CH3COOH
2CH3COOH
CH3CH2OH��CH3COOH![]() CH3COOCH2CH3��H2O��
CH3COOCH2CH3��H2O��
��4��![]() ����5��D��
����5��D�� ��F ��CH3CH2OOCCH2COCH3���м����1Ϊ��
��F ��CH3CH2OOCCH2COCH3���м����1Ϊ�� ���м����2��
������2��
����������1��A�Ƿ�����������C�Ľṹ��ʽ���Ƴ�A��Ӧ��6��̼ԭ�ӣ���AΪ�����ṹ��ʽΪ��![]() ��A��B����ȡ����Ӧ�����룭NO2�����B�й���������������ȷ�𰸣�
��A��B����ȡ����Ӧ�����룭NO2�����B�й���������������ȷ�𰸣�![]() ����������2������C��D�ķ���ʽ��C��D���������ǻ�ȡ��������λ�ã������ķ�Ӧ��ȡ����Ӧ����ȷ�𰸣�ȡ����Ӧ����3��E��������һ�������������Ҵ���F����E���������������Ҵ���ȡ�������������ò����Ҵ�ͨ�������Ʊ����ᣬ�ٺ���һ�����Ҵ�ͨ��������Ӧ�õ�������������ȷ�𰸣�2CH3CH2OH+O2
����������2������C��D�ķ���ʽ��C��D���������ǻ�ȡ��������λ�ã������ķ�Ӧ��ȡ����Ӧ����ȷ�𰸣�ȡ����Ӧ����3��E��������һ�������������Ҵ���F����E���������������Ҵ���ȡ�������������ò����Ҵ�ͨ�������Ʊ����ᣬ�ٺ���һ�����Ҵ�ͨ��������Ӧ�õ�������������ȷ�𰸣�2CH3CH2OH+O2 ![]() 2CH3CHO+2H2O 2CH3CHO+O2
2CH3CHO+2H2O 2CH3CHO+O2 ![]() 2CH3COOH CH3CH2OH��CH3COOH
2CH3COOH CH3CH2OH��CH3COOH![]() CH3COOCH2CH3��H2O����4�������֪ת��������ԭ���غ㣬�Ƴ�F�ķ���ʽΪC6H10O3��������Ϣ���Լ��Ǽ��㶹�صĽṹ��ʽ���Ƴ�F�Ľṹ��ʽΪCH3CH2OOCCH2COCH3��������
CH3COOCH2CH3��H2O����4�������֪ת��������ԭ���غ㣬�Ƴ�F�ķ���ʽΪC6H10O3��������Ϣ���Լ��Ǽ��㶹�صĽṹ��ʽ���Ƴ�F�Ľṹ��ʽΪCH3CH2OOCCH2COCH3��������![]() ��������
��������![]() ����ȷ�𰸣�
����ȷ�𰸣�![]() ����5�������ǻ��㶹�صĽṹ��ʽ���Լ���2���ķ�����C��D�İ������ǻ��ֱ��ڱ����ļ�λ���ֱ�Ϊ
����5�������ǻ��㶹�صĽṹ��ʽ���Լ���2���ķ�����C��D�İ������ǻ��ֱ��ڱ����ļ�λ���ֱ�Ϊ ��
�� ��F�Ľṹ��ʽΪCH3CH2OOCCH2COCH3��F��D������֪��һ����Ӧ�����м����1���м����1�Ľṹ��ʽΪ��
��F�Ľṹ��ʽΪCH3CH2OOCCH2COCH3��F��D������֪��һ����Ӧ�����м����1���м����1�Ľṹ��ʽΪ�� ���ٷ�����֪���ķ�Ӧ�����м����2��
���ٷ�����֪���ķ�Ӧ�����м����2�� ��Ȼ������ȥ��Ӧ��ˮ�����Ǽ��㶹�ء���ȷ�𰸣�D��
��Ȼ������ȥ��Ӧ��ˮ�����Ǽ��㶹�ء���ȷ�𰸣�D�� ��F ��CH3CH2OOCCH2COCH3���м����1Ϊ��
��F ��CH3CH2OOCCH2COCH3���м����1Ϊ�� ���м����2��
������2��

 �����Ƹ���ʦ����ϵ�д�
�����Ƹ���ʦ����ϵ�д� ��ͨ����ͬ����ϰ��ϵ�д�
��ͨ����ͬ����ϰ��ϵ�д� ����С����ͬ������ϵ�д�
����С����ͬ������ϵ�д�


