��Ŀ����
ʳ�κ�ҵ���ξ��й㷺����;����֪��ҵ������Ҫ�ɷ�ΪNaNO2����ۿ���ʳ�β�����ζ��NaNO2�������Ժͻ�ԭ�ԣ�����ֽ�ų�NO2��
��1�������Լ��ɼ���ҵ���κ�ʳ�ε���________��
a��H2O��������������b�������ữ�������� c������
��2��NaNO2������ᷴӦ����������ΪNO�������ӷ���ʽΪ______________��
��3��ij������Һ�к���2%��5%��NaNO2��ֱ���ŷŻ����ˮ��Ⱦ�����������������е�ijһ�־���ʹNaNO2�еĵ�ת��Ϊ�Կ�������Ⱦ�����壬��������________��
a��NaCl������b��NH4Cl������c��ŨH2SO4
��4���ȼҵͨ����ⱥ��ʳ��ˮ�������Ӧ�IJ����д�����ʳ��ˮ�����ӷ���ʽ______________________________________��
���ڱ�״��������3.36 L H2������Һ�����ɵ�OH�������ʵ���Ϊ____��
��1��bc
��2��2NO2-��2I����4H��=I2��2NO����2H2O
��3��b
��4��2Cl����2H2O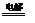 Cl2����H2����2OH����0.3 mol
Cl2����H2����2OH����0.3 mol
����

��ҵ��ˮ�г�����һ������Cr2O72����CrO42�������ǻ�����༰��̬ϵͳ�����ܴ���˺���������д������÷��Ĺ�������Ϊ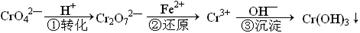
���еڢٲ�����ƽ�⣺2CrO42������ɫ��+2H+ Cr2O72������ɫ��+H2O
Cr2O72������ɫ��+H2O
��1����ƽ����ϵ��pH=2������Һ�� ɫ��
��2����˵���ڢٲ���Ӧ��ƽ��״̬���� ����ѡ���ţ�
| A��Cr2O72����CrO42����Ũ����ͬ | B��v��(Cr2O72��) ="2v" ��(CrO42��) |
| C����Һ����ɫ���� | D����Һ��pHֵ���� |
��4����Cr2(SO4)3��Һ�У��μ�NaOH����pH��4.6ʱ����ʼ����Cr(OH)3����������pH�����ߣ��������࣬����pH��13ʱ��������ʧ����������ɫ��[Cr(OH)4]�����ӡ���ƽ���ϵ���£�
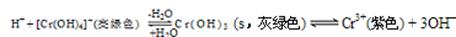
����0.05mol��L��1��Cr2(SO4)3��Һ50mL�У���������0.6 mol��L��1��NaOH��Һ����ַ�Ӧ����Һ�пɹ۲쵽������Ϊ ����Һ������Ũ���ɴ�С��˳��Ϊ ��
��5����Na[Cr(OH)4]��Na2Cr2O7��Ϻ����Һ�м���H2SO4�ữ����Ԫ���� ��ʽ���ڣ���д���ӷ��ţ���
�������ȣ�ClO2����Ŀǰ�����Ϲ��ϵĵ��Ĵ���Ч����������������һ�ֻ���ɫ�����壬������ˮ��
��.��1��ClO2����KClO3��H2SO4���ڵ���������Na2SO3��Ӧ�Ƶá���÷�Ӧ�����������뻹ԭ��������ʵ���֮����________��
��.ʵ����Ҳ����NH4Cl�����ᡢNaClO2���������ƣ�Ϊԭ���Ʊ�ClO2�����������£�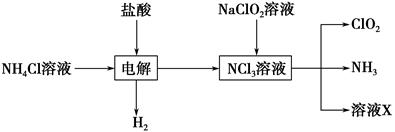
��2��д�����ʱ������Ӧ�Ļ�ѧ����ʽ��________________________________��
��3����ȥClO2�е�NH3��ѡ�õ��Լ���________��������ţ�
| A������ʳ��ˮ | B����ʯ�� |
| C��Ũ���� | D��ˮ |
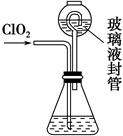
��װ���в���Һ��ܵ�������______________________________________��
����д��������������������⻯����Һ��Ӧ�����ӷ���ʽ__________________________��
�۵ζ��յ��������_______________________________________________��
�ܲ��ͨ��ClO2������m��ClO2����________�����ú�c��V�Ĵ���ʽ��ʾ��
��5����ClO2������������ˮ��pHΪ5.5��6.5��������һ���������岻���������������ClO2-��2001���ҹ��������涨������ˮ��ClO2-����Ӧ������0.2 mg��L��1��������ˮ��ClO2-�ĺ������꣬�������м���������ij��ԭ�����÷�Ӧ������������________���ѧʽ�����䷢����Ӧ�����ӷ���ʽΪ_________________________________________________________________��
����ع㷺Ӧ���ڻ�϶�������ϵͳ���缫������Ni��OH��2��̿�ۡ���������Ϳ�����������Ƴɣ����ڵ��ʹ�ú�缫���϶Ի�����Σ����ij��ȤС��Ըõ�ص缫���Ͻ�����Դ�����о�������Ƴ����ʵ������ͼ
��֪����NiCl2������ˮ��Fe3+��������Ni2+
��ij�¶���һЩ�������������Ksp����������������pH�����ʾ��
| M��OH��n | Ksp | pH | |
| ��ʼ���� | ������ȫ | ||
| Al��OH��3 | 1.9��10-33 | 3.43 | 4.19 |
| Fe��OH��3 | 3.8��10-38 | 2.53 | 2.94 |
| Ni��OH��2 | 1.6��10-14 | 7.60 | 9.75 |
�ش��������⣺
��1�����ݱ������жϲ������������������ �ͳ����� ���ѧʽ������pH1 pH2�������������=�����������������ֳ������������� ��
A��pH��ֽ B��ʯ��ָʾ�� C��pH��
��2����֪�ܽ�ȣ�NiC2O4��NiC2O4?H2O��NiC2O4?2H2O����Ӧ�۵Ļ�ѧ����ʽ�� ���ڢ۲���Ӧ���˳���ʱ��Ҫ�IJ��������� ��������ʱ������Һ�����������ǣ���ʵ������ĽǶȸ������ֿ��ܵ�ԭ�� �� ��
��3������������Ӧ����������EΪ ����֤��������Լ�Ϊ ��
��4����д����Ӧ�����ӷ���ʽ_____________________________________________
ij��ҵ��ˮ�����±��е�ijЩ���ӣ��Ҹ������ӵ����ʵ���Ũ����ȣ���Ϊ0.1 mol/L(����ֵ����ˮ�ĵ��뼰���ӵ�ˮ��)��
| ������ | K����Ag����Mg2����Cu2����Al3����NH4+ |
| ������ | Cl����CO32����NO3����SO42����SiO32����I�� |
��ͬѧ��̽����ˮ����ɣ�����������ʵ�飺
��.ȡ����ɫ��Һ5 mL���μ�һ�ΰ�ˮ�г������ɣ��������������ӡ�
��.�ò�˿պȡ��Һ���ڻ��������գ�����ɫ�ܲ����۲죬����ɫ���档
��.��ȡ��Һ����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ��
��.��������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɡ�
���ƶϣ�
(1)�ɢ��жϣ���Һ��һ�������е���������____________��
(2)���м�������������ɫ��������ӷ���ʽ��
___________________________________________________________��
(3)��ͬѧ����ȷ��ԭ��Һ��������������________����������________�����ݴ��Ʋ�ԭ��ҺӦ�ó�_______________________________________________�ԣ�ԭ����_________________________________(�������ӷ���ʽ˵��)��
(4)��ȡ100 mLԭ��Һ������������NaOH��Һ���˹������漰�����ӷ���ʽΪ__________________________________________________________��
��ַ�Ӧ����ˣ�ϴ�ӣ����ճ��������أ��õ��Ĺ�������Ϊ________g��
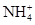 ��Cu2+��Ba2+��Al3+��Ag+��Fe3+�������ӿ�����Cl-��
��Cu2+��Ba2+��Al3+��Ag+��Fe3+�������ӿ�����Cl-��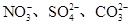 ����֪��
����֪��