��Ŀ����
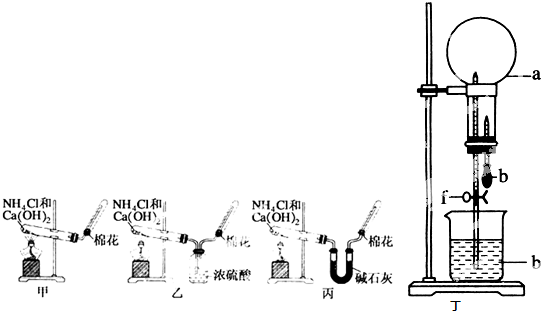
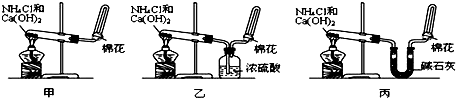
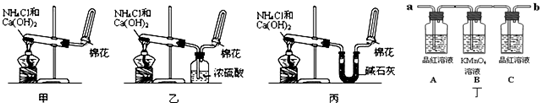
��10�֣��ס��ҡ�����λͬѧ�ֱ�����������ʵ��װ�ü���ѧҩƷ�����м�ʯ��Ϊ�����������ƺ���ʯ�ҵĻ�����ȡ�������������̽�������ش��������⣺
��1����λͬѧ��ȡ�����Ļ�ѧ����ʽΪ��_______________________
��2����λͬѧ���������ſ������ռ���������������ˮ������ԭ����_________
A������������ˮ B��������������ˮ
C�������ܶȱȿ����� D�������ܶȱȿ���С
E�������ܶȱ�ˮ�� F�������ܶȱ�ˮС
��3����λͬѧ������װ����ȡ����ʱ,������ͬѧû���ռ����������ռ�������������Ҫԭ����____________________ (�û�ѧ����ʽ��ʾ)��
��4�����鰱���Ƿ��ռ����ķ�����_________
A���ŵ��а����ݳ�
B������������
C����ʪ��ĺ�ɫʯ����ֽ���Թܿڼ��飬������ֽ����
D����ʪ�����ɫʯ����ֽ���Թܿڼ��飬������ֽ���
��5����λͬѧ����Ϊ���ǵ�ʵ��װ��Ҳ�����ڼ���̼����粒�����ȡ�����İ��������ж��ܹ��ﵽʵ��Ŀ�ĵ���___________(��ס������ҡ�����)��
��1��2NH4Cl+Ca��OH��2====CaCl2+2NH3��+2H2O
��2��BD
��3��2NH3+H2SO4===��NH4��2SO4
��4��C
��5����
����:

 �ܿ�����ĩ��̾�ϵ�д�
�ܿ�����ĩ��̾�ϵ�д�