��Ŀ����
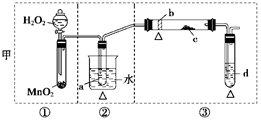
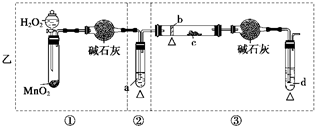
��10�֣��ڳ����£�Fe��ˮ������Ӧ�����ڸ����£�Fe��ˮ�����ɷ�����Ӧ��Ӧ������װ�ã���Ӳ�ʲ������з��뻹ԭ���ۺ�ʯ���Ļ������ȣ���ͨ��ˮ�������Ϳ�����ɸ�����"Fe��ˮ������Ӧ��ʵ��"��

��ش��ʵ���е����⡣
��1��д���÷�Ӧ�Ļ�ѧ����ʽ ��
��2��ʵ��ǰ���������װ�ý��еIJ����� ��
��3��Ӳ�ʲ�������ʯ������Ҫ������ ��
��4���������ʢװ�����ʿ�����
��5����ȼ�ƾ��ƺ;ƾ���Ƶ��Ⱥ�˳����

��ش��ʵ���е����⡣
��1��д���÷�Ӧ�Ļ�ѧ����ʽ ��
��2��ʵ��ǰ���������װ�ý��еIJ����� ��
��3��Ӳ�ʲ�������ʯ������Ҫ������ ��
��4���������ʢװ�����ʿ�����
��5����ȼ�ƾ��ƺ;ƾ���Ƶ��Ⱥ�˳����
(1) 3Fe + 4H2O(g)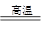 Fe3O4 + 4H2
Fe3O4 + 4H2
(2) ���������
(3) ��������ˮ�����ĽӴ����
(4) ��ʯ��
(5) �ȵ�ȼ�ƾ����ٵ�ȼ�ƾ����
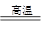 Fe3O4 + 4H2
Fe3O4 + 4H2(2) ���������
(3) ��������ˮ�����ĽӴ����
(4) ��ʯ��
(5) �ȵ�ȼ�ƾ����ٵ�ȼ�ƾ����
��������ˮ�����ķ�Ӧ
ˮ��������ƿ�е�ˮ���ƾ��Ƽ������ã�ͬʱΪ������Ӧ��ĽӴ�������ں��ʲ�������ʢװʯ����
Ϊ�˷�ֹ���۵�������ͬʱΪ�˷�ֹ�����������ı�ը�����ڷ�Ӧ��ʼʱ��Ӧ���ȵ�ƾ��ƣ������ܹ��Ȳ���ˮ�����ž��������ڵĿ�������ʵ�����ʱ��Ӧ����Ϩ��ƾ���ƣ�
ˮ��������ƿ�е�ˮ���ƾ��Ƽ������ã�ͬʱΪ������Ӧ��ĽӴ�������ں��ʲ�������ʢװʯ����
Ϊ�˷�ֹ���۵�������ͬʱΪ�˷�ֹ�����������ı�ը�����ڷ�Ӧ��ʼʱ��Ӧ���ȵ�ƾ��ƣ������ܹ��Ȳ���ˮ�����ž��������ڵĿ�������ʵ�����ʱ��Ӧ����Ϩ��ƾ���ƣ�

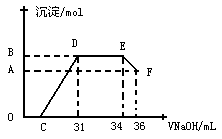
��ϰ��ϵ�д�
�����Ŀ
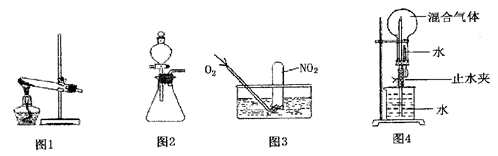



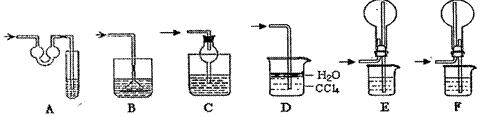
 չ���о������и��л�ѧ�̲��жԡ����ȷ�Ӧ��������������������������Ӧ�ų��������ȣ�������ҫ�۵Ĺ�â������ֽ©�����²����մ���������������ɳ�С������ġ���ѧ
չ���о������и��л�ѧ�̲��жԡ����ȷ�Ӧ��������������������������Ӧ�ų��������ȣ�������ҫ�۵Ĺ�â������ֽ©�����²����մ���������������ɳ�С������ġ���ѧ �ֲᡷ֪��Al��Al2O3��Fe��Fe2O3�۵㡢�е��������£�
�ֲᡷ֪��Al��Al2O3��Fe��Fe2O3�۵㡢�е��������£�