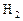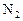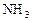��Ŀ����
��������Ч���ý�����������еĽ������仯�����л�ԭ��������������������ֽ������ϣ���һ�����ڹ�ҵ�ϳ�Ϊ������ұ����
��1����HgOΪԭ��ұ��Hg��ұ�������� ��
��2�����ȷ���ұ�������ķ���֮һ��д�����ȷ�����(Fe2O3)�Ļ�ѧ����ʽ�� ���������ȷ�Ӧ��ʵ������ǣ�
��
��3��ijЩ�����������ĩ��������һ�������¿��Է������ȷ�Ӧ�����з�Ӧ���ʣ��ԣ����¶ȣ�T���Ĺ�ϵʾ��ͼ�������ȷ�Ӧ��ӽ����ǣ� ������ĸ����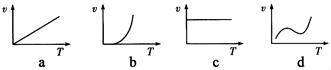
��1����HgOΪԭ��ұ��Hg��ұ�������� ��
��2�����ȷ���ұ�������ķ���֮һ��д�����ȷ�����(Fe2O3)�Ļ�ѧ����ʽ�� ���������ȷ�Ӧ��ʵ������ǣ�
��
��3��ijЩ�����������ĩ��������һ�������¿��Է������ȷ�Ӧ�����з�Ӧ���ʣ��ԣ����¶ȣ�T���Ĺ�ϵʾ��ͼ�������ȷ�Ӧ��ӽ����ǣ� ������ĸ����

��ÿ��2�֣���8�֣���1�����ȷֽⷨ
��2��Al��Fe2O3 Al2O3 ��2Fe�� ������KClO3������Mg���������ȼ��
Al2O3 ��2Fe�� ������KClO3������Mg���������ȼ��
��3��b
��2��Al��Fe2O3
 Al2O3 ��2Fe�� ������KClO3������Mg���������ȼ��
Al2O3 ��2Fe�� ������KClO3������Mg���������ȼ�� ��3��b
��

��ϰ��ϵ�д�
 �Ͻ�ƽ��У����ϵ�д�
�Ͻ�ƽ��У����ϵ�д�
�����Ŀ
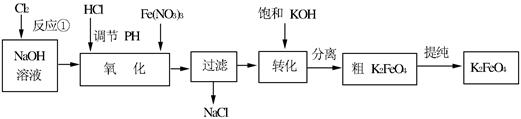
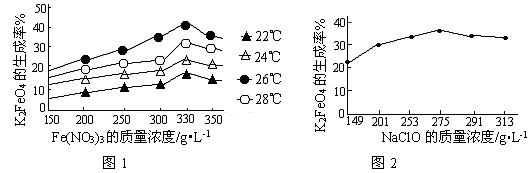
 4Fe(OH)3+8OH��+3O2�����ڡ��ᴿ��K2FeO4�в����ؽᾧ��ϴ�ӡ����º�ɵķ�������ϴ�Ӽ����ѡ��_______��Һ������ţ���
4Fe(OH)3+8OH��+3O2�����ڡ��ᴿ��K2FeO4�в����ؽᾧ��ϴ�ӡ����º�ɵķ�������ϴ�Ӽ����ѡ��_______��Һ������ţ���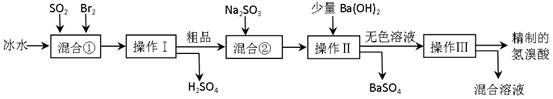
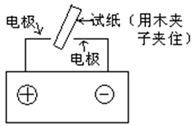
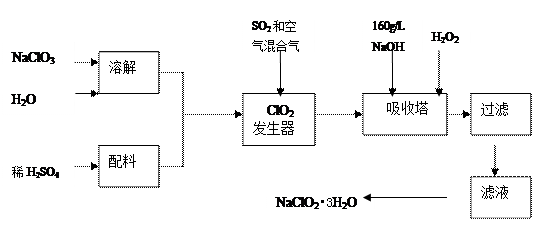


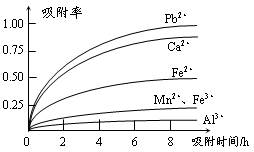
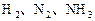 ��Ӧ��ƽ��ʱ������Ũ������һ���ǣ� ��
��Ӧ��ƽ��ʱ������Ũ������һ���ǣ� ��