��Ŀ����
����Ŀ������(�Ҷ���)������ԭ���ͳ����������ڽ������⡢֯��Ư��ϡ��������һ���Ʊ�����(��2���ᾧˮ)�Ĺ����������ң�

�ش��������⣺
��1��CO��NaOH��һ�������ºϳɼ����ơ������Ƽ�������Ļ�ѧ��Ӧ����ʽ�ֱ�Ϊ��__________________��_______________��
��2�����Ʊ������������ι��˲��������˲���������Һ��___________��������________�����˲���������Һ��____________��____________��������_______��
��3�����չ�������������Ŀ����______________��
��4�����˽�������������ֱ���������ữ�Ʊ����ᡣ�÷�����ȱ���Dz�Ʒ���������к��е�������Ҫ��____________________��
��5���ᾧˮ�ϲ����Ʒ�Ĵ����ø�����ط��ⶨ��
���������Ʒ0.250 g����ˮ����0.0500 mol��L-1������KMnO4��Һ�ζ�����dz�ۺ�ɫ�����ʣ�����KMnO4��Һ15.00 mL����Ӧ�����ӷ���ʽΪ__________________��
��ʽ����ó�Ʒ�Ĵ���____________________________��
���𰸡�CO��NaOH![]() HCOONa 2HCOONa
HCOONa 2HCOONa![]() Na2C2O4��H2�� NaOH��Һ CaC2O4 H2C2O4��Һ H2SO4��Һ CaSO4 �ֱ�ѭ�������������ƺ�����(���ͳɱ�)����С��Ⱦ Na2SO4 5C2O42��+2MnO4��+16H��==2Mn2��+8H2O+10CO2��
Na2C2O4��H2�� NaOH��Һ CaC2O4 H2C2O4��Һ H2SO4��Һ CaSO4 �ֱ�ѭ�������������ƺ�����(���ͳɱ�)����С��Ⱦ Na2SO4 5C2O42��+2MnO4��+16H��==2Mn2��+8H2O+10CO2�� 
��������
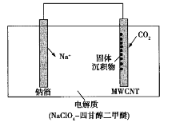
��������ͼ֪��һ�������£�һ����̼���������Ʒ�Ӧ���ɼ����ƣ���Ӧ����ʽΪ��CO��NaOH![]() HCOONa�����������£��������������ɲ����ƣ���ԭ���غ��֪���������ɣ���Ӧ����ʽΪ2HCOONa
HCOONa�����������£��������������ɲ����ƣ���ԭ���غ��֪���������ɣ���Ӧ����ʽΪ2HCOONa![]() Na2C2O4��H2�������������������Ʒ�Ӧ�õ���������������ƣ���Һ��NaOH��Һ��Ũ����ѭ�����ã�����ΪCa C2O4������������ᷴӦ�õ������������ƣ�������ΪCaSO4����ҺBΪ��������Һ���پ����ᾧ������õ������ƾ��壬ĸҺ�к�������Ͳ��ᣬ��ѭ�����á�
Na2C2O4��H2�������������������Ʒ�Ӧ�õ���������������ƣ���Һ��NaOH��Һ��Ũ����ѭ�����ã�����ΪCa C2O4������������ᷴӦ�õ������������ƣ�������ΪCaSO4����ҺBΪ��������Һ���پ����ᾧ������õ������ƾ��壬ĸҺ�к�������Ͳ��ᣬ��ѭ�����á�
��1�����ݹ�������ͼ��֪��CO��NaOH��һ�������ºϳɼ����Ʒ���ʽΪCO��NaOH![]() HCOONa�������Ƽ��������ͬʱ���в��������ɣ���˷�Ӧ�Ļ�ѧ��Ӧ����ʽΪ2HCOONa
HCOONa�������Ƽ��������ͬʱ���в��������ɣ���˷�Ӧ�Ļ�ѧ��Ӧ����ʽΪ2HCOONa![]() Na2C2O4��H2����
Na2C2O4��H2����
��2�����ݷ�Ӧԭ�����ܽ����Լ�ʾ��ͼ��֪�����˲���������Һ��NaOH��Һ��������CaC2O4�����˲���������Һ��H2C2O4��Һ��H2SO4��Һ��������CaSO4��
��3�����ݹ��չ��̿�֪������������ѭ�������������ƣ�����������ѭ���������ᣬ�������ԭ�ϵ������ʣ��ֽ����˳ɱ�����С��Ⱦ��
��4�������������IJ���Ϊ�����ƣ�ֱ���������ữ�����ɲ���������ƣ����к��е�������Ҫ��Na2SO4��
��5���ڲⶨ�����У��������Ϊ������������Ϊ��ԭ������Ӧ�����ӷ���ʽ5C2O42- + 2MnO4- + 16H+= 2Mn2++ 8H2O + 10CO2�������ݷ���ʽ�ɵù�ϵʽ��
5H2C2O4��2H2O ~ 2KMnO4
5 2
n 0.05mol/L��15.0��10-3L
���n��H2C2O4��2H2O��=1.875��10-3mol
��m��H2C2O4��2H2O��=1.875��10-3mol��126g/mol=0.236g
���Գ�Ʒ�Ĵ�����=![]() ��100%=94.5%��
��100%=94.5%��

 ����һ������ܼƻ�ϵ�д�
����һ������ܼƻ�ϵ�д�����Ŀ����֪25��ʱ����(H2C2O4)�ĵ��볣��ΪK1=5.0��10-2��K2=5.4��10-5������Ƶ�Ksp=4.0��10-8��̼��Ƶ�Ksp=2.5��10-9����ͬ�¶���ˮ�����ӻ��������±���
t/�� | 0 | 10 | 20 | 25 | 40 | 50 | 90 | 100 |
Kw/10-14 | 0.134 | 0.292 | 0.681 | 1.00 | 2.92 | 5.57 | 38.0 | 55.0 |
(1)�����½�0.2 mol/L��KOH��Һ20 mL��0.2 mol/L�IJ�����Һ20 mL��Ϻ���Һ�����ԣ����Ϻ���Һ�и�����Ũ�ȵĴ�С˳��Ϊ__________________________________��
(2)��֪������ʹ���Ը��������Һ(ϡ�����ữ)��ɫ��������ƿ�з���20.00mLδ֪Ũ�ȵIJ��ᣬ��0.1mol/L���������Һ�ζ���������ζ��յ�ʱ����������VmL���������Һ�����������ʵ���Ũ��Ϊ__________mol/L��
(3)90��ʱ����0.005 mol/L������������Һ20 mL��0.0012 mol/L�IJ�����Һ20 mL��ϣ���Ϻ���Һ��pH=___________��
(4)25��ʱ����20 mL����Ƶı�����Һ����μ���8.0��10-4 mol/L��̼�����Һ10 mL���ܷ����������___________(����������������)��