��Ŀ����
����ݽ̲��ϵ�ʵ�顰����������Ӧ��������������⡣
��1��ȡһ���̶���Ľ����ƣ���ȥ�����㣩������ֽ����������ú�ͣ�����ʯ�����ϣ��þƾ����ȡ������۳���״ʱ����ʢ�������ļ���ƿѸ�ٵ������Ƶ��Ϸ���������ѧ��֪ʶ������ʵ�������Щȱ�㣿
��____________________________________________________��
��____________________________________________________��
��________________����������2����
��2��ijͬѧ�������ϴ��ڵ�ȱ��Ľ�ʵ��װ�ã���ͼ��ʾ��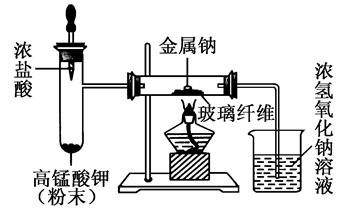
ʵ�鲽�裺
a��ȡ�̶�������ƣ�����ֽ���ɱ����ú�ͣ���ȥ�����㣬���벣�����У���ͼʾ��װ��������
b�������μ�Ũ���ᣬ�������ҷ�Ӧ����������
c�����������г�������ɫ����ʱ���ټ����ƣ����ۻ���ȼ�ա�
�ٹ۲쵽��ʵ�������У����μ�Ũ������Թ��в���________ɫ���壻�ƾ���ȼ�գ������________ɫ����________���ɣ���Ӧ�����ܱ��Ϲ۲쵽��________���ɡ�
�ڸĽ����ʵ���ŵ㣺a.________��b.________��c.______�����ٻش�2������
��д��Na��Cl2��ȼ�յĻ�ѧ����ʽ________________�����õ���ʽ��ʾ�����γɹ���________________��
��1����NaԤ���ڿ����м��ȣ����������������Ӱ��Na��Cl2��ȼ��
��Ԥ���ռ���Cl2�ڽ���ʵ��ʱ���ܲ��������
��ʵ������л������Ⱦ
��2���ٻ��̡��ơ����̡���ɫ����
��a.����ʵ���������������һ��Ũ�Ⱥʹ��ȣ����ⷢ������Ӧ��b����ȫ�ɿ�����Ⱦ�١�c�����Ա������ڼ���ʱ����Na2O2
��2Na��Cl2 2NaCl
2NaCl 
����

��ʵ���Ҳⶨ̼������̼�����ƵĻ�����У�̼���Ƶ���������[�÷���w(Na2CO3)��ʾ]����ȡ�˻����5.lg������ˮ�У����250mL��Һ��
a��(10��)����һ����������w(Na2CO3)���û�ѧ��Ӧ��HCO3����CO32����ȫת��Ϊ��������ȡ�������������ɴ˼���������w (Na2CO3)��
��1����ȡ100 mL���ƺõ���Һ���ձ��У��μ�����������������Һ��HCO3����CO32����ȫת��Ϊ������Ӧѡ���Լ���___________ �����ţ���
| A��CaCl2 | B��MgSO4 | C����NaCI | D��Ba(OH)2 |
��3�����ˣ���ȡ����������˲�������Ҫ�IJ���������________________________��
��4��ϴ�ӳ���������ϴ�ӳ����IJ���_____________________________��
��5�������֣���ȡ����������Ϊ9.8g���ɴ˼���w(Na2CO3)������˲��У�����δ�����־ͳ���������w (Na2CO3)________________����ƫ���ƫС����Ӱ�죩��
b�����������ζ�����w(Na2CO3)��ȡ25.00 mL���ƺõ���Һ������ƿ�У��μ�2�η�̪�Լ���ҡ�ȣ���0.2000 mol/L��������еζ����յ㡣�ظ��˲���2�Σ�������������ƽ��ֵΪ20.00 mL�� [��֪���͵�̼����ҺPHΪ3.9]
��1����ȡ25.00 mL���ƺõ���Һ��Ӧѡ��_______________��������ɡ�
��2���жϵζ��յ��������_____________________���˹����з�Ӧ�����ӷ���ʽΪ__________________________________________________��
��3���˷����w(Na2CO3)=________%��������λС����
������������������ͭ���Ǻ�ɫ��ĩ�����������ϡ�ijУһ��ѧʵ��С��ͨ��ʵ����̽��һ��ɫ��ĩ��Fe2O3��Cu2O����ߵĻ���̽���������£�
�������ϣ�Cu2O��һ�ּ������������ϡ��������Cu��CuSO4���ڿ����м�������CuO��
�������
����1����ɫ��ĩ��Fe2O3
����2����ɫ��ĩ��Cu2O
����3����ɫ��ĩ��Fe2O3��Cu2O�Ļ����
���̽��ʵ��
ȡ������ĩ��������ϡ�����У���������Һ���ٵμ�KSCN�Լ���
��1��������1��������ʵ��������_____________________________________________��
��2�����μ�KSCN�Լ�����Һ�����ɫ����֤��ԭ�����ĩ��һ����������������������Ϊ����˵��������________�������������(����д����Ӧ����ʽ)____________
________________________________________________________________________��
��3���������ĩ��ȫ�ܽ�������ڣ��μ�KSCN�Լ�ʱ��Һ�����ɫ����֤��ԭ�����ĩ��________��д��������Ӧ�����ӷ���ʽ________________________________��
̽������
��ʵ�������ȷ����ɫ��ĩΪFe2O3��Cu2O�Ļ���
��4��ʵ��С�����ü��ȷ��ⶨCu2O������������ȡa g�����ĩ�ڿ����г�ּ��ȣ����������ٱ仯ʱ����������Ϊb g(b��a)����������Cu2O����������Ϊ________��
��5��ʵ��С�������øú�ɫ��ĩ��ȡ�ϴ����ĵ���(CuSO4��5H2O)�����������ϵ�֪������Һ��ͨ��������Һ������Զ�ʹCu2����Fe2����Fe3���ֱ����ɳ�����pH���£�
| ���� | Cu(OH)2 | Fe(OH)2 | Fe(OH)3 |
| ��ʼ����pH | 6.0 | 7.5 | 1.4 |
| ������ȫpH | 13 | 14 | 3.7 |
ʵ�����������Լ��ɹ�ѡ��A.��ˮ��B��H2O2��C��NaOH��D��Cu2(OH)2CO3
ʵ��С���������ʵ�鷽����
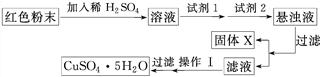
�Իش�
���Լ�1Ϊ________(����ĸ����ͬ)���Լ�2Ϊ________��
�ڹ���X�Ļ�ѧʽΪ____________________________________________________��
�۲�����Ϊ___________________________________________________________��
Ϊ��̽��SO2��Na2O2�ķ�Ӧ�Ƿ�������CO2��Na2O2�ķ�Ӧ����ͬѧ�������ͼ��ʾ��ʵ��װ�ã��ش��������⣺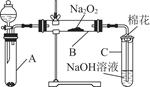
��1���ƿ������������ǵ�ľ������C�Թܿڣ�δ��ľ����ȼ����ͬѧ�����ΪSO2��Na2O2�ķ�Ӧ��ͬ��CO2���밴��ͬѧ�Ĺ۵�д����Ӧ�Ļ�ѧ����ʽ ��
��2����ͬѧ��Ϊ���۷�Ӧԭ����Σ����ն���O2��������ͬѧ�������� ��������ͬѧ�Ĺ۵㣬��װ�������ĸĽ���
��
��3������Na2O2��ȫ��Ӧ����Ӧ��Bװ���й�������������ǣ���Na2SO3����Na2SO4����Na2SO3��Na2SO4��
�����ʵ�鷽�����飬д��ʵ�鲽���Լ�Ԥ������ͽ��ۣ�����±���
��ѡ�Լ���2 mol��L��1 HCl��Һ��1 mol��L��1 HNO3��Һ��1 mol��L��1 BaCl��Һ��1 mol��L��1 Ba��NO3��2��Һ��0.01 mol��L��1 KMnO4������Һ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡB�е�����������Ʒ���Թ��У��μ���������ˮ���ܽ⣬Ȼ��ȡ��������Һ�ֱ����ڢ��Թ��� | ������ȫ�ܽ� |
| ����2�������Թ��м��� ���ٵμ� | �� |
| ��֤���������к�Na2SO4 | |
| ����3�������Թ��� | |
| | �� �� |
| ��֤������������Na2SO3���� | |
| | |
| ��˵����������û��Na2SO3�� | |
��4�����������������ƺ����IJⶨ��
��ȡa g���������Ƴ�100 mL��Һ��ȡ10.00 mL����Һ����ƿ�У����뼸�ε�����Һ��ָʾ������0.010 0 mol��L��1��ˮ���еζ����ζ��յ�����Ϊ ����¼���ݣ��ظ��ζ�2�Σ�ƽ�����ĵ�ˮ20.00 mL��
�ڼ��㣺���������������Ƶ���������Ϊ ��


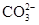 ������ȫ���ܷ�ʹ���Ȼ�����Һ�����Ȼ�����Һ��_______ ����ܡ�����ԭ���ǣ�_____________________________________��
������ȫ���ܷ�ʹ���Ȼ�����Һ�����Ȼ�����Һ��_______ ����ܡ�����ԭ���ǣ�_____________________________________��
