��Ŀ����
����Ŀ�����Ĺ̶�����������淢չ������Ҫ���壬��ѧ��һֱ�����ڡ��˹��̵������·����о���
(1)����ֲ��ĸ���������һ�̵ֹ�ø�����ڳ����½������еĵ���ת��Ϊ������������ĺ������������˵���������________(����)��
a���̵�ø��һ�ֵ�����
b���̵�ø�ܽ��̵���Ӧ�������������
c���̵�ø��ʹ�̵���Ӧ��ƽ�������ƶ�
d���̵�ø�ܼӿ�̵���Ӧ������
(2)�ϳɰ���Ŀǰ����Ҫ�Ĺ̵���������ͼΪ�ϳɰ���ӦN2(g)��3H2(g)![]() 2NH3(g)�ڲ�ͬ�¶Ⱥ�ѹǿ��ʹ����ͬ���������£���ʼʱ���������������֮��Ϊ1��3ʱ��ƽ�������а�����������ı仯��������ͼ��
2NH3(g)�ڲ�ͬ�¶Ⱥ�ѹǿ��ʹ����ͬ���������£���ʼʱ���������������֮��Ϊ1��3ʱ��ƽ�������а�����������ı仯��������ͼ��
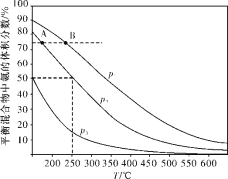
��A��B��ƽ��״̬��Ƚϣ�ǰ�߽ϴ����___________(����)��
a��ƽ�ⳣ�� b��ƽ�������ƽ��Ħ������
c��������ת���� d���ӷ�Ӧ��ʼ���ﵽƽ��״̬����Ҫ��ʱ��
��ͼ��p1��p2��p3�ɴ�С��˳��Ϊ_____________________��
����250 �桢p2�£�H2��ת����Ϊ_____________%(�������С�����һλ)��
(3)�ݱ������ڳ��¡���ѹ�����������£�N2�ڴ���(��������Fe2O3��TiO2)������ˮ������Ӧ�����ɵ���Ҫ����ΪNH3��һ�ֵ��ʡ���֪����H2(g)��![]() O2(g)===H2O(l)����H1����285.6 kJ/mol����N2(g)��3H2(g)
O2(g)===H2O(l)����H1����285.6 kJ/mol����N2(g)��3H2(g)![]() 2NH3(g)����H2����92.2 kJ/mol��д���������µ�����ˮ��Ӧ���Ȼ�ѧ����ʽ��________________��
2NH3(g)����H2����92.2 kJ/mol��д���������µ�����ˮ��Ӧ���Ȼ�ѧ����ʽ��________________��
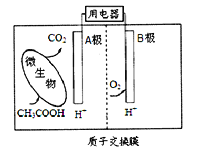
(4)��ͼΪһ�ֳ��³�ѹ��ˮ��Һ�е绯ѧ�ϳɰ���ʵ��װ��ʾ��ͼ����ݴ�д����������Ӧ�ĵ缫��Ӧʽ��_________________________________��
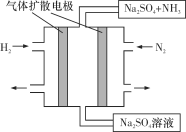
���𰸡�cadp1>p2>p366.72N2(g)��6H2O(l)===4NH3(g)��3O2(g) ��H����1529.2kJ/molN2��6e����6H2O===2NH3��6OH��(��N2��6e����6H��===2NH3)
��������
��1��a��ø�����ڵ����ʣ����Թ̵�ø��һ�ֵ����ʣ���a˵����ȷ��
b���̵�ø���˴��������ã����Թ̵�ø�ܽ��̵���Ӧ���������������ʹ��Ӧ�ڳ����¾Ϳ��Խ��У���b˵����ȷ��
c������ֻ�ܸı䷴Ӧ���ʣ�����Ӱ�컯ѧƽ�⣬���Թ̵�ø����ʹ�̵���Ӧ��ƽ�������ƶ�����c˵������
d�������ܹ��ӿ췴Ӧ���ʣ����Թ̵�ø�ܼӿ�̵���Ӧ�����ʣ���d˵����ȷ��
�ʴ�Ϊ��c��
��2����a��ƽ�ⳣ��ֻ���¶�Ӱ�죬����ͼ���֪��A����¶�С��B���ϳɰ��ķ�ӦΪ���ȷ�Ӧ���¶����ߣ�ƽ�����������ƶ���ƽ�ⳣ����С������A��ƽ�ⳣ������B����a��ȷ��
b��A��B������ƽ��״̬ʱ�����������������ȣ��������������������Ҳ��ȣ����������ƽ��Ħ��������A=B����b����
c��������ת���ʣ�����A��B�ﵽƽ��״̬ʱ����ֵĺ�����ȫ��ͬ������������ת���ʣ�A=B����c����
d��B��ѹǿ���¶ȶ�����A����B�ķ�Ӧ���ʴ���A������B���ȴﵽƽ��״̬�����ӷ�Ӧ��ʼ����ƽ��״̬����Ҫ��ʱ�䣺A��B����d��ȷ��
�ʴ�Ϊ��ad��
����ͬ�¶��£�ѹǿ����ƽ�����������ƶ���������������������������������ϵΪ��p1��p2��p3����ѹǿ��СΪ��p1��p2��p3���ʴ�Ϊ��p1��p2��p3��
����250����1.0��104kPa�£���ʼʱ�����������������Ϊ1��3���������������Ϊ50%���赪����ת����Ϊx����
N2+3H2![]() 2NH3
2NH3
��ʼ 1 3 0
ת�� x 3x 2x
ƽ�� 1-x 3-3x 2x
����![]() =50%�����x=
=50%�����x=![]() ����������ת����Ϊ��
����������ת����Ϊ��![]() =x=
=x=![]() ��100%��66.7%��
��100%��66.7%��
�ʴ�Ϊ��66.7��
��3����H2��g��+![]() O2��g���TH2O��1����Hl�T-285.6kJ/mol��N2��g��+3H2��g���T2NH3��g����H2�T-92.2kJ/mol��������ˮ��Ӧ�Ļ�ѧ����ʽΪ��2N2��g��+6H2O��l��=4NH3��g��+3O2��g�������ݸ�˹���ɣ�����2-����6�ɵã�2N2��g��+6H2O��l��=4NH3��g��+3O2��g����H=2��H2-6��Hl�T+1529.2kJ/mol���ʴ�Ϊ��2N2��g��+6H2O��l��=4NH3��g��+3O2��g����H=+1529.2kJ/mol��
O2��g���TH2O��1����Hl�T-285.6kJ/mol��N2��g��+3H2��g���T2NH3��g����H2�T-92.2kJ/mol��������ˮ��Ӧ�Ļ�ѧ����ʽΪ��2N2��g��+6H2O��l��=4NH3��g��+3O2��g�������ݸ�˹���ɣ�����2-����6�ɵã�2N2��g��+6H2O��l��=4NH3��g��+3O2��g����H=2��H2-6��Hl�T+1529.2kJ/mol���ʴ�Ϊ��2N2��g��+6H2O��l��=4NH3��g��+3O2��g����H=+1529.2kJ/mol��
��4������ͼʾ��֪����������ʧȥ���ӷ���������Ӧ���ɰ������缫��ӦʽΪ��N2-6e-+6H2O=2NH3+6OH-����N2-6e-+6H+=2NH3�����ʴ�Ϊ��N2-6e-+6H2O=2NH3+6OH-����N2-6e-+6H+=2NH3����

����Ŀ���о���Ԫ�ؼ��仯��������ʾ�����Ҫ���塣
��. ��������ȼ�ջ����������������Ҫ�ɷ���SO2��CO2��N2��O2 ��ij�о���ѧϰС����ʵ������������װ���Ʊ�ģ�����������ⶨ������SO2�����������
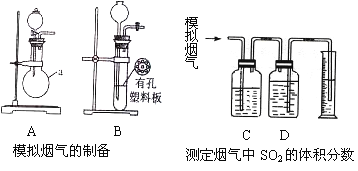
��һ��ģ���������Ʊ�
��1����Aװ����SO2����ѧ��Ӧ����ʽΪ___________________________��
��2����Bװ����CO2��ʹ�ø�װ���ŵ����___________________________��
��3�����Ƶõ������������ֻ�ϣ����ģ���������ں���ʵ�顣
�������ⶨ������SO2���������
��4��������ģ����������ͨ��C��Dװ�ã�����C��D��ʢ�е�ҩƷ�ֱ���__________��__________��������ţ�
��KMnO4��Һ ������NaHSO3��Һ ������Na2CO3��Һ �ܱ���NaHCO3��Һ
��5����ģ������������ΪamL/min����t1���Ӻ����Ͳ��Һ�����ΪVmL����SO2���������____________________��
��.ij��ѧ��ȤС���ͬѧ������ͼ��ʾʵ��װ�ý���ʵ��(ͼ��a��b��c��ʾֹˮ��) ��
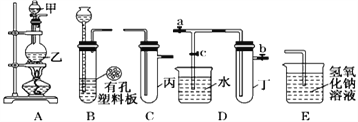
��1��A��C��E�������װ�ÿ�������ȡCl2��������ص�����ʵ�顣�����ڱ��м�������ˮ�������Ƶ���ˮ����������ˮ����ʵ�飬ʵ����������������£�
ʵ����� | �� �� | �� �� |
��ˮ�м���̼�����Ʒ�ĩ | ����ɫ���ݲ��� | ������ˮ��Ӧ�IJ���������� |
ʵ��Ľ����Ƿ����__________����ѡ�������������˵������______________________��
������������װ�����һ����ʵ����֤Cl-��S2-�Ļ�ԭ��ǿ��,��ס��ҡ�����ʢ�ŵ��Լ��Ǽ�__________________ ��__________________ ��__________________
��2��B��D��Eװ����������B��ʢװŨ�����ͭƬ(�����п����ϰ���)�����Ƶò�����NO2�й�ʵ�顣����Dװ����֤NO2��ˮ�ķ�Ӧ�����������Ϊ���ȹر�ֹˮ��________���ٴ�ֹˮ��________��ʹ�ձ��е�ˮ�����Թܶ��IJ����ǣ�___________________________________ʹ�Թ��е������ݳ���NO2��ˮ�Ӵ��������ձ��е�ˮ�����Թܶ���