��Ŀ����
10��ij������ˮ�Ļ������ĩ�г�����̿���⣬�����ܺ����������ĵ��ʺ����ǵ�ijһ�������ijУʵ��С��ͨ��ʵ��ȷ���û�����ĩ�ijɷ֣�ʵ�鲽�裺
��ȡm g�����ĩ��һ�ձ��У���������NaOHϡ��Һ��������ų������˵õ�����A����ɫ��ҺB��
�ڽ��������ù���Aϴ�Ӻ�ֳ����ݣ�һ�ݼ���CuSO4��Һ�������Ա仯����һ�ݼ�������ϡ���ᣬ���˵õ�����C���ػ�ɫ��ҺD��
��ش��������⣺
��1��д��������з�����Ӧ�����ӷ���ʽ2Al+2OH-+6H2O=2[Al��OH��4]-+3H2����
��2������C��һ����������������Ʒ�����Ӧ�Ļ�ѧ����ʽΪ2CO2+2Na2O2=2Na2CO3+O2��
��3�����ػ�ɫ��ҺD�м������ۣ�������Ӧ�����ӷ���ʽΪ2Fe3++Fe=3Fe2+��
��4��������ʵ����Եó����ۣ�������ĩ��һ������̼��Al���ѧʽ����ͬ��������Ԫ�ص����ʿ�����Fe2O3��Fe3O4����Ҫȷ������Ԫ�ص����ʣ�����Ҫ����һ��ʵ�飬��ʵ�鷽��Ϊȡ��������KMnO4��Һ���Թ��У����������ػ�ɫ��ҺD������ɫ��Һ��ɫ����˵��ԭ����ﺬ��Fe3O4������ɫ��Һ����ɫ����˵��ԭ������к���Fe2O3��
���� �ٹ����ĩ��һ���ձ��У���������NaOHϡ��Һ�������������֤��һ�����н����������˵õ�����A����ɫ��ҺB����ҺBΪNaAlO2��Һ��
�ڽ��������ù���Aϴ�Ӻ�ֳ����ݣ�һ�ݼ���CuSO4��Һ�������Ա仯��֤��һ������������һ�ݼ�������ϡ���ᣬ���˵õ�����C���ػ�ɫ��ҺD���ƶϹ���CΪ̼�ۣ��ػ�ɫ��ҺDΪ��������Һ��֤��һ��������������������������
������������Ϸ�ĩ�� һ����������̼�����������һ���������������ƶϳ������ʷ����ش�
��1���ٷ�Ӧ�����ӷ���ʽ����������������Һ�ķ�Ӧ����ƫ�����ƺ�������
��2������C��һ����������������Ʒ�����Ӧ�Ƕ�����̼�������Ʒ�Ӧ����̼���ƺ�������
��3����Ӧ�����������ӷ�Ӧ�����������ӣ�
��4��������ʵ����Ի�ý��ۣ�������ĩ��һ������̼�������������������������������������������Ҫȷ������Ԫ�ص����ʣ���Ҫ�����ܽ⣬�����������Ӿ��л�ԭ�ԣ�ʹ���������Һ��ɫ���ʵ����֤��
��� �⣺ij������ˮ�Ļ�Ϸ�ĩ�г�����̿���⣬�����ܺ����������ĵ��ʺ����ǵ�ijһ�������
�ٹ����ĩ��һ���ձ��У���������NaOHϡ��Һ�������������֤��һ�����н����������˵õ�����A����ɫ��ҺB����ҺBΪNaAlO2��Һ��
�ڽ��������ù���Aϴ�Ӻ�ֳ����ݣ�һ�ݼ���CuSO4��Һ�������Ա仯��֤��һ������������һ�ݼ�������ϡ���ᣬ���˵õ�����C���ػ�ɫ��ҺD���ƶϹ���CΪ̼�ۣ��ػ�ɫ��ҺDΪ��������Һ��֤��һ���������������������������ƶ�Ϊ����Ϸ�ĩ��һ����������̼�����������һ����������
��1���ٷ�Ӧ�����ӷ���ʽ����������������Һ�ķ�Ӧ�����ӷ���ʽΪ��2Al+2OH-+2H2O=2AlO2-+3H2����
�ʴ�Ϊ��2Al+2OH-+2H2O=2AlO2-+3H2����
��2������C��һ����������������Ʒ�����Ӧ�Ƕ�����̼�������Ʒ�Ӧ����̼���ƺ���������Ӧ�Ļ�ѧ����ʽΪ��2CO2+2Na2O2=2Na2CO3+O2��
�ʴ�Ϊ��2CO2+2Na2O2=2Na2CO3+O2��
��3�����ػ�ɫ��ҺD��Fe2��SO4��3���м������۷�Ӧ�����ӷ���ʽΪ��2Fe3++Fe=3Fe2+���ʴ�Ϊ��2Fe3++Fe=3Fe2+��
��4��������ʵ����Ի�ý��ۣ�������ĩ��һ������̼�������������������������������������������Ҫȷ������Ԫ�ص����ʣ��������һ��ʵ�飬��ʵ�鷽��Ϊ��ȡ�����������Թ��У�����ϡ������ܣ�ȡ�ϲ���Һ������������Һ������ɫ֤����������������������ɫ֤������������������������Ϊ��������
�ʴ�Ϊ��Al��Fe2O3��Fe3O4��ȡ�����������Թ��У�����ϡ������ܣ�ȡ�ϲ���Һ������������Һ������ɫ֤����������������������ɫ֤������������������������Ϊ��������
���� ���⿼�������ʼ��鷽����Ӧ�ã��������ʵķ����жϣ���Ҫ�����ʼ���ķ���������Ӧ�ã���ѧ����ʽ�����ӷ���ʽ����д���������ʼ���ķ�����ƣ���Ŀ�Ѷ��еȣ�

| A�� | ԭ��������ͬ | B�� | ��ԭ������ͬ | C�� | ��������ͬ | D�� | ��ԭ������ͬ |
| A�� | �ᳫ��̼���� | B�� | ������չ˽�ҳ� | ||
| C�� | �������̫���ܡ����� | D�� | ú��������Һ������ |
| A�� | ���ȷ�Ӧ | B�� | ���ȷ�Ӧ | ||
| C�� | �ؼ���Ӧ | D�� | ����ЧӦ��������ЧӦ |
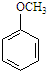

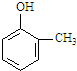
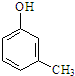
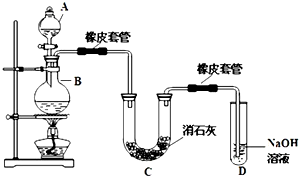 ��仯ѧ���������о����̿���Ҫ�ɷ���MnO2 ���Ĺ����У�������Ũ����������ȣ�������һ�ֻ���ɫ���̼���������ζ������----������ijѧ��ʹ����һԭ���������ͼ��ʾ��ʵ��װ�ã����������Ƶõ������볱ʪ����ʯ�ҷ�Ӧ��ȡ����Ư�ۣ�����һ�����ȷ�Ӧ�����ݴ˻ش��������⣺
��仯ѧ���������о����̿���Ҫ�ɷ���MnO2 ���Ĺ����У�������Ũ����������ȣ�������һ�ֻ���ɫ���̼���������ζ������----������ijѧ��ʹ����һԭ���������ͼ��ʾ��ʵ��װ�ã����������Ƶõ������볱ʪ����ʯ�ҷ�Ӧ��ȡ����Ư�ۣ�����һ�����ȷ�Ӧ�����ݴ˻ش��������⣺