��Ŀ����
��ͼ��ʾ���ϳɰ�������ʾʵ��(�г���������ʡ��)����Y�ιܵ�һ����Zn����ϡH2SO4��Ӧ��ȡH2����һ����NaNO2�����NH4Cl������Һ��Ӧ��ȡN2��N2��H2��Ϻ�ͨ����ԭ�������ϳ�NH3���ٽ�����������ͨ���̪��Һ�У�����̪��Һ��죬��˵�������˰�����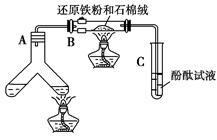
ij����С��ͨ���������ϺͶ��ʵ�飬�õ���������Ϣ��
��Ϣһ��NaNO2����ͱ���NH4Cl��Һ��ϼ��ȵĹ����з������·�Ӧ��
��NaNO2��NH4Cl NH4NO2��NaCl
NH4NO2��NaCl
��NH4NO2 NH3����HNO2
NH3����HNO2
��2HNO2 N2O3����H2O
N2O3����H2O
��2NH3��N2O3 2N2��3H2O
2N2��3H2O
��Ϣ�����������ϣ���ͬ����ȵ�N2��H2�����������ͬʵ�������ºϳɰ���ʹ��̪��Һ�������Ҫ��ʱ�����£�
| N2��H2������� | 5��1 | 3��1 | 1��1 | 1��3 | 1��5 |
| ��̪���ɫ����ʱ��/min | 8��9 | 7��8 | 6��7 | 3��4 | 9��10 |
�ݴ˻ش��������⣺
(1)Y�ι������з�����Ӧ�����ӷ���ʽ________________________��
(2)��������ʯ�����ϵ�Ŀ����_________________________________
(3)����С���ͬѧ����Ϊ����ʵ���м�ʹ��̪���Ҳ����˵��N2��H2��Ӧ�ϳ���NH3���ó��˽��۵�������________________________��
���������һ����ʵ����֤�������____________________���������һ���⣬����ѡ����ͼ�е�________װ��������ԭװ���е�________��________֮�䡣
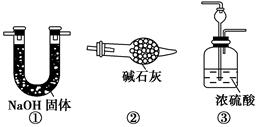
(4)������ʵ������У�Ϊ����۲쵽��̪��Һ����ʵ������Ӧ�ÿ���N2��H2�������Ϊ________�Ƚ����ˣ�����װ�û�����ʵ�ִ�Ŀ�ģ�ԭ����______________________________________��
(5)ʵ�������ͨ���Թ�C�е�����ɷ���________��
(1)Zn��2H��=Zn2����H2��
(2)����������������ĽӴ������ʹ��Ӧ���еø���
(3)�ӷֲ���Ӧ��֪������N2�Ĺ����У��п���ֱ�Ӳ�������������ϼ��Ȳ���������ֱ��ͨ���̪��Һ������Һ��죬��˵�����ɳ���������˵�����ɲ��������ۡ�A��B
(4)1��3��������ͨ��B��N2��H2�������
(5)NH3��N2��H2
����

 �Ƹ�360�ȶ����ܾ�ϵ�д�
�Ƹ�360�ȶ����ܾ�ϵ�д� ���⿼����Ԫ���Ծ�ϵ�д�
���⿼����Ԫ���Ծ�ϵ�д� ��У���˳�̾�ϵ�д�
��У���˳�̾�ϵ�д� ��У���һ��ͨϵ�д�
��У���һ��ͨϵ�д����л�ѧʵ�������ܹ��ﵽĿ�ĵ���
| A��Ϊ����KCl��AlCl3��MgCl2��Һ���ֱ���������Һ�еμ�NaOH��Һ������ |
| B������ȥ��������Һ�е�NaCl���ֲ��ı������ʣ��ɼ�������BaCl2��Һ����� |
| C������ˮ��pH�����ò�����պȡ��ˮ����pH��ֽ�ϣ������ɫ��ͱ���ɫ���Ƚ� |
| D��Ϊ��֤����¯���к����������ɽ���¯��ͨ�����ȵ�����ͭ��ĩ������ɫ��ĩ�Ƿ���ɫ |
��ʽ̼����A������θҩ������ɿɱ�ʾΪAl2Mg6(OH)x(CO3)y��zH2O��ijУ��ѧ��ȤС�����ⶨ�仯ѧʽ��ʵ��������£�
ʵ��I����ȡһ��������A�����ȷֽ������ء�
ʵ���ȡһ��������A���������ᷴӦ����������CO2�����������
�ɹ�ѡ���������ҩƷ��ͼ��ʾ��������Һ��ѡ6mol/LHCl��6mol/LH2SO4�������Լ���ѡ����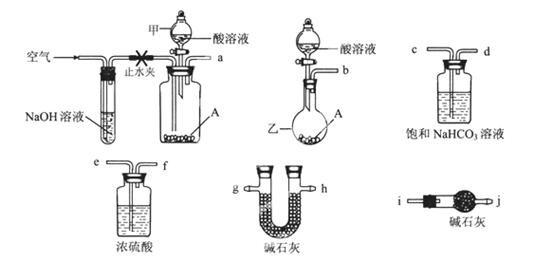
�ش��������⣺
(1)�����ҵ�����Ϊ________��
(2)��ѡ���Ҫ��װ�����ʵ��II,��ȷ������˳��Ϊ________ (�����������ýӿ���ĸ��ʾ����ѡ�õ�����Һ��________��
(3)�������������ʵ��I������ʵ��II��������A��ȫ��Ӧ��������Һ�еμ������İ�ˮ��������ֽ���ˣ�������ˮϴ�ӷ�Ӧ����2?3�Σ���ϴ��Һ���ˣ�ϴ�ӳ���2?3�Σ������ų�������ֽ�ŵ������м��ȷֽ������ء��жϳ�����ϴ�Ӹɾ��ķ�����_________________,ʵ����δ���ø÷�����ԭ���Dz�����ʵ����Ƶ�________ԭ������ĸ��ţ���
| A����ѧ�� | B����ȫ�� | C�������� | D����Լ�� |
(5)ͨ��ʵ��I��������ݣ�������ɵ�A��Ħ������Ϊ602.0g. mol-1��ʵ��II�гƵ���ƷA������Ϊ9.030g������������ȫ��Ӧ��CO2����װ������0.660g,��A �Ļ�ѧʽΪ________��
��1��С���������о��¶ȶԷ�Ӧ���ʵ�Ӱ�족ʵ��ʱ����ȡ����ֻ�Թܣ�������4mL 0.01mol��L��KMnO4������Һ��2mL 0.1mol/L H2C2O4���Ҷ��ᣩ��Һ����A�Թ�������ˮ�У�B�Թ�������ˮ�У���¼��Һ��ɫ�����ʱ�䡣
����Ҫ�� ���ữKMnO4��Һ����ɫ����ʱ��tA tB���>������=����<������
��д���÷�Ӧ�����ӷ���ʽ ��
��2��ʵ������ƿ������ɳ���Ҷ�����Ʒ��С�����������Ӧ��ԭ�����ⶨ�京�����������Ϊ��
������250 mL��Һ��ȷ����5.0g�Ҷ�����Ʒ�����250mL��Һ��
�ڵζ���ȷ��ȡ25.00 mL������Һ����ƿ�У����������ữ����0.1000 mol��L��1 KMnO4��Һװ�� �����ʽ����ʽ�����ζ��ܣ����еζ�������
��ʵ���з��֣��յ�������KMnO4��Һʱ����ҺѸ�ٱ���Ϻ�ɫ������ƿҡ��һ��ʱ����Ϻ�ɫ������ʧ���ټ����μ�ʱ���Ϻ�ɫ�ͺܿ���ɫ�ˡ������ԭ��
����____
��֤���ﵽ�ζ��յ㡣
�ۼ��㣺���ظ���������2�Σ���¼ʵ���������¡�
| ��� | �ζ�ǰ������mL�� | �ζ��������mL�� |
| 1 | 0.00 | 20.10 |
| 2 | 1.00 | 20.90 |
| 3 | 1.10 | 21.10 |
������KMnO4��Һ��ƽ�����Ϊ mL����֪H2C2O4����Է�������Ϊ90�������Ʒ�Ĵ���Ϊ ��
�������������в����ᵼ�²ⶨ���ƫ�ߵ��� ��
A��δ�ñ�Ũ�ȵ�����KMnO4��Һ��ϴ�ζ���
B���ζ�ǰ��ƿ������ˮ
C���ζ�ǰ�ζ��ܼ��첿�������ݣ��ζ���������ʧ
D����С�Ľ���������KMnO4��Һ������ƿ��
E���۲����ʱ���ζ�ǰ���ӣ��ζ�����
 NH2COONH4(s) ��H��0
NH2COONH4(s) ��H��0 

 ����)������Һ�м�������Ca(OH)2��Һ��Ȼ���ټ�������CH3COOH��Һ���۲�����
����)������Һ�м�������Ca(OH)2��Һ��Ȼ���ټ�������CH3COOH��Һ���۲�����