��Ŀ����
10���Ӻ�ˮ����ȡ�����Ҫ������������1�����ữ�ĺ�ˮ��ͨ��������������ʹ������ת��Ϊ�����壺2NaBr+Cl2�TBr2+2NaCl
��2�����������ڵ���Һ��ͨ��������������2HBr+Cl2�T2HCl+Br2
��3��������ˮŨ���������ὫŨ���ĺ�ˮ�ữ
��4�������嵥�ʵ�ˮ��Һ��ͨ�������ˮ���������嵥�ʴ���ʢ�ж���������Һ�����������Դﵽ������Ŀ�ģ�Br2+SO2+2H2O�T2HBr+H2SO4
��5�������Ȼ�̼������ȡ�������ڵ���Һ�е��嵥�ʣ�
��ȷ��˳���ǣ�������
| A�� | ��1����2����3����4����5�� | B�� | ��3����1����4����2����5�� | C�� | ��5����1����3����2����4�� | D�� | ��5����4����3����2����1�� |
���� ��ˮ������Ũ����ˮ���������ὫŨ���ĺ�ˮ�ữ���ữ�ĺ�ˮ��ͨ���������������������ӷ���������ԭ��Ӧ�����嵥�ʣ����嵥�ʵ�ˮ��Һ��ͨ�������ˮ���������嵥�ʴ���ʢ�����������Һ�б���ԭΪ�廯�⣬��������������Һ��ͨ����������������������Ϊ�嵥�ʣ�������ȡ��Һ������ȡ�嵥�ʣ�
��� �⣺�Ӻ�ˮ����ȡ�����Ҫ�������̵�˳��Ϊ��
��3����ˮ������Ũ����ˮ���������ὫŨ���ĺ�ˮ�ữ��
��1���ữ�ĺ�ˮ��ͨ���������������������ӷ���������ԭ��Ӧ�����嵥�ʣ�2Br-+Cl2=Br2+2Cl-��
��4�����嵥�ʵ�ˮ��Һ��ͨ�������ˮ���������嵥�ʴ���ʢ�����������Һ�б���ԭΪ�廯�⣬Br2+SO2+2H2O=H2SO4+2HBr��
��2����������������Һ��ͨ����������������������Ϊ�嵥�ʣ�2HBr+Cl2=Br2+2HCl��
��5��������ȡ��Һ������ȡ�嵥�ʣ�
������˳��Ϊ����3����1����4����2����5����
��ѡB��
���� ���⿼���˺�ˮ����Ĺ�ҵ�������̺��Լ�ѡ��ע��ԭ���ķ����жϣ���ѧ����ʽ��д�ǹؼ�����Ŀ�ϼ�

��ϰ��ϵ�д�
 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д�
�����Ŀ
18����A��B��C���ֶ�����Ԫ�أ���֪AԪ�ص�ԭ��������������������Ӳ�����BԪ�ص�ԭ������������������Ӳ�����2����CԪ�ص�ԭ������������������Ӳ�����3����������Ԫ����ɵĻ�����Ļ�ѧʽ�������ǣ�������
| A�� | A3BC4 | B�� | A2��BC4��3 | C�� | A2BC3 | D�� | A2BC4 |
5�����й��ڶ�±��Ԫ�����ʵıȽ�˵������ȷ���ǣ�������
| A�� | ���ϵ����⻯����۷е������� | |
| B�� | ���ϵ��µ��ʵĻ�ԭ������ | |
| C�� | ���ϵ�������������ˮ������������ | |
| D�� | ���ϵ���ԭ�ӵõ��ӵ���������ǿ |
15�����з�Ӧ�У����ڼӳɷ�Ӧ���ǣ�������
| A�� | ������ˮ��ϣ����ã���ˮ����ɫ | |
| B�� | ��ϩͨ�������{�������Һ�У���Һ��ɫ | |
| C�� | ��ϩͨ������ķջ�̼��Һ�У���Һ��ɫ | |
| D�� | ������������Ϲ��գ�������ɫ��dz |
2�����в��ֶ�����Ԫ�ص����ʻ�ԭ�ӽṹ���±���
��1���û�ѧ����ش��������⣺
��AԪ�������ڱ��е�λ�õ������ڢ�A�壻
��BԪ��ԭ�ӽṹʾ��ͼ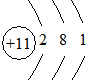 ��
��
��C���ʷ��ӵĵ���ʽ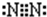 ���õ���ʽ��ʾA��BԪ����ɵĻ�������γɹ���
���õ���ʽ��ʾA��BԪ����ɵĻ�������γɹ���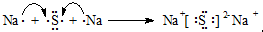 ��
��
��DԪ������Ȼ�������ֺ��أ���ԭ�ӷ��ű�ʾ��������Ϊ20�ĺ���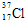 ��
��
��2��Ԫ��D��Ԫ��A��ȣ��ǽ����Խ�ǿ����Cl����Ԫ�ط��ű�ʾ�������б�������֤����һ��ʵ����bde����ѡ����ţ���
a��������D�ĵ��ʺ�A�ĵ���״̬��ͬ
b��D���⻯���A���⻯���ȶ�
c��һ��������D��A�ĵ��ʶ������Ʒ�Ӧ
d��A����ۺ�������������D����ۺ�����
e��D��������A���⻯�ﷴӦ����A����
��3��̽Ѱ���ʵ����ʲ�������ѧϰ����Ҫ����֮һ��A��B��C��D����Ԫ�ص�����������ˮ�����л�ѧ�������Բ�ͬ���������ֵ���NaOH��д��ѧʽ����
��4��X����A��B��C��D����Ԫ���е�ij��Ԫ����ɵĵ��ʣ��ܾ���ͼ��ʾ�Ĺ���ת��ΪW������������ȥ����
X$\stackrel{O_{2}}{��}$Y$\stackrel{O_{2}}{��}$Z$\stackrel{H_{2}O}{��}$W
����Y���д̼�����ζ����ɫ���壬��Yͨ��BaCl2��Һ�У�Ȼ��μ�����H2O2��Һ���а�ɫ�������ɣ��˰�ɫ�����Ļ�ѧʽΪ��BaSO4�����ɸð�ɫ�����Ļ�ѧ����ʽΪBaCl2+SO2+H2O2=BaSO4��+2HCl����Ba2++SO2+H2O2=BaSO4��+2H+����
����Z�Ǻ���ɫ���壬��Z��W�ķ�Ӧ���������뻹ԭ�����ʵ���֮��Ϊ��1��2��
| Ԫ�ر�� | Ԫ�����ʻ�ԭ�ӽṹ |
| A | ���������Ӳ㣬K��M�������֮�͵���L������� |
| B | �������н�������ǿ |
| C | �����µ���Ϊ˫ԭ�ӷ��ӣ��⻯���ˮ��Һ�ʼ��� |
| D | Ԫ�����������+7�� |
��AԪ�������ڱ��е�λ�õ������ڢ�A�壻
��BԪ��ԭ�ӽṹʾ��ͼ
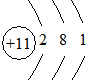 ��
����C���ʷ��ӵĵ���ʽ
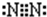 ���õ���ʽ��ʾA��BԪ����ɵĻ�������γɹ���
���õ���ʽ��ʾA��BԪ����ɵĻ�������γɹ��� ��
����DԪ������Ȼ�������ֺ��أ���ԭ�ӷ��ű�ʾ��������Ϊ20�ĺ���
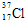 ��
����2��Ԫ��D��Ԫ��A��ȣ��ǽ����Խ�ǿ����Cl����Ԫ�ط��ű�ʾ�������б�������֤����һ��ʵ����bde����ѡ����ţ���
a��������D�ĵ��ʺ�A�ĵ���״̬��ͬ
b��D���⻯���A���⻯���ȶ�
c��һ��������D��A�ĵ��ʶ������Ʒ�Ӧ
d��A����ۺ�������������D����ۺ�����
e��D��������A���⻯�ﷴӦ����A����
��3��̽Ѱ���ʵ����ʲ�������ѧϰ����Ҫ����֮һ��A��B��C��D����Ԫ�ص�����������ˮ�����л�ѧ�������Բ�ͬ���������ֵ���NaOH��д��ѧʽ����
��4��X����A��B��C��D����Ԫ���е�ij��Ԫ����ɵĵ��ʣ��ܾ���ͼ��ʾ�Ĺ���ת��ΪW������������ȥ����
X$\stackrel{O_{2}}{��}$Y$\stackrel{O_{2}}{��}$Z$\stackrel{H_{2}O}{��}$W
����Y���д̼�����ζ����ɫ���壬��Yͨ��BaCl2��Һ�У�Ȼ��μ�����H2O2��Һ���а�ɫ�������ɣ��˰�ɫ�����Ļ�ѧʽΪ��BaSO4�����ɸð�ɫ�����Ļ�ѧ����ʽΪBaCl2+SO2+H2O2=BaSO4��+2HCl����Ba2++SO2+H2O2=BaSO4��+2H+����
����Z�Ǻ���ɫ���壬��Z��W�ķ�Ӧ���������뻹ԭ�����ʵ���֮��Ϊ��1��2��
20���������ӷ���ʽ��д����ȷ���ǣ�������
| A�� | NH4HCO3���ڹ�����NaOH��Һ�У�HCO3-+OH-�TCO32-+H2O | |
| B�� | �Ȼ�������Һ��ϡ�����ϣ�3Fe2++4H++NO3-�T3Fe3++2H2O+NO�� | |
| C�� | AlCl3��Һ�м��������İ�ˮ��Al3++4OH-�TAlO2-+2H2O | |
| D�� | NO2����ˮ��3NO2+H2O�T2H++2NO3-+NO |
 ij��ȤС���������ͼ��ʾ��ʵ��װ�ã��ȿ�������ȡ���壬�ֿ�������֤���ʵ����ʣ�
ij��ȤС���������ͼ��ʾ��ʵ��װ�ã��ȿ�������ȡ���壬�ֿ�������֤���ʵ����ʣ� ��1����ʵ��������ȡ��ϩ�����¶ȹ��߶�ʹ�Ҵ���ŨH2SO4��Ӧ����������SO2�����������ͼ��ʾʵ����ȷ�����������������C2H4��SO2�� ����ϩ����ȡװ���ԣ�
��1����ʵ��������ȡ��ϩ�����¶ȹ��߶�ʹ�Ҵ���ŨH2SO4��Ӧ����������SO2�����������ͼ��ʾʵ����ȷ�����������������C2H4��SO2�� ����ϩ����ȡװ���ԣ�