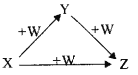
��Ŀ����
����Ŀ��ij����С�����þ��CO2�ķ�Ӧ���ʵ��̽��þ��NO2�ķ�Ӧ��
��ͬѧ�Ʋ������MgO��N2��
��ͬѧ�Ʋ�������MgO��N2�⣬�����л����ܺ���Y��
��С��ͬѧ���������װ��̽��þ��NO2��Ӧ�Ĺ��������ⶨ����ɡ�
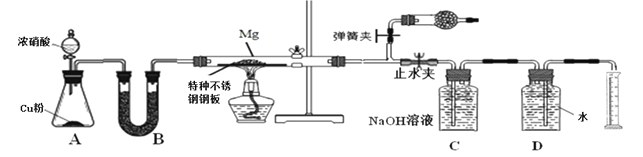
��1��ʵ�鿪ʼʱ���ȹر�ֹˮ�к���ɼУ��ٴ�Һ©����������Ӳ�ʲ����ܳ�������ɫ�����ֹˮ�У��رյ��ɼУ�����ȼ�ƾ��ơ���������Ŀ����___________________________________________________________
��2��װ��B�е��Լ�����ѡ��________
A��Ũ���� B����ˮ�Ȼ��� C������������ D����ʯ��
��3��װ��C��ʢװ����������Һ�������ǣ�___________________________________
��4��Ϊ��֤�������������ȷ�ԣ���ȡ��Ͳ�̶�ʱӦע��������Ǣ�����ָ��������ٶ�������_______________________________����______________________________��
��5��ʵ���������ͬѧ�ǽ��������ȡ����ˮ��Ӧ�������д̼�����ζ�������������������ʹʪ���ʯ����ֽ������˵������ͬѧ�Ʋ���ȷ����д��Y��ˮ��Ӧ�Ļ�ѧ����ʽ__________________________________
��6������ʼ����þ������Ϊ3.6 g����������NO2�г�ַ�Ӧ�� ���ռ���N2���Ϊ448mL (��״��)���������MgO��������_________
���𰸡� �ų�װ���п���������������� BC ����ʣ��NO2 ������Ͳ��Һ����D��ˮƽ ������Һ�尼Һ������ Mg3N2+H2O=3Mg(OH)2��+2NH3�� 4.8g
��������(1)ʵ�鿪ʼʱ���ȹر�ֹˮ�к���ɼУ��ٴ�Һ©����������Ӳ�ʲ����ܳ�������ɫ�����ֹˮ�У��رյ��ɼУ�����ȼ�ƾ��ơ������������ų�װ���п���������������ţ��ʴ�Ϊ���ų�װ���п���������������ţ�
(2)װ��A��Ũ������ͭ��Ӧ���ɶ������������ɵĶ��������л���ˮ������װ��B�е��Լ���Ҫ��ȥ���е�ˮ����������ѡ����ˮ�Ȼ��ƺ��������������Թ�����������ѡBC��
(3)���������ܹ���Ⱦ������������ˮ��Ӧ����һ��������Ӱ�쵪��������IJⶨ��װ��C��ʢװ����������Һ��������Ϊ��Ӧ��NO2���ʴ�Ϊ������ʣ���NO2��
(4)Ϊ��֤�������������ȷ�ԣ���ȡ��Ͳ�̶�ʱӦע��������ǣ�������ָ��������ٶ������ڵ�����Ͳ��Һ����D��ˮƽ����������Һ�尼Һ�����У��ʴ�Ϊ��������Ͳ��Һ����D��ˮƽ��������Һ�尼Һ�����У�
(5)ʵ�������ͬѧ�ǽ��������ȡ����ˮ��Ӧ�������д̼�����ζ�������������������ʹʪ���ʯ����ֽ������˵�����ɵ�����Ϊ���������������к��е�Ԫ�أ����ݲ��뷴Ӧ������Ϊþ�Ͷ��������������жϹ������ΪMg3N2��Mg3N2��ˮ��Ӧ�Ļ�ѧ����ʽΪMg3N2+H2O=3Mg(OH)2��+2NH3�����ʴ�Ϊ��Mg3N2+H2O=3Mg(OH)2��+2NH3����
(6)þ�����ʵ���=![]() =0.15mol����Ӧ��������þ��þ��ʧȥ����0.3mol����״���£�N2���Ϊ448mL�����ʵ���=
=0.15mol����Ӧ��������þ��þ��ʧȥ����0.3mol����״���£�N2���Ϊ448mL�����ʵ���=![]() =0.02mol���õ��˵���0.02mol��8=0.16mol�����ݵ�ʧ�����غ㣬����Mg3N2
=0.02mol���õ��˵���0.02mol��8=0.16mol�����ݵ�ʧ�����غ㣬����Mg3N2![]() mol=0.01mol������þԭ���غ㣬���ɵ�����þΪ
mol=0.01mol������þԭ���غ㣬���ɵ�����þΪ![]() mol=0.12mol������Ϊ0.12mol��40g/mol=4.8g���ʴ�Ϊ��4.8g��
mol=0.12mol������Ϊ0.12mol��40g/mol=4.8g���ʴ�Ϊ��4.8g��

����Ŀ������ˮ��ɽ���ǽ�ɽ��ɽ����������,��ɫ��չ����̬������Ϊ�й�չʾ�������һ���¡���Ƭ��������β������ɴ�����Ⱦ����Ҫԭ��֮һ,���ٵ����������ڴ����е��ŷ��ǻ�����������Ҫ����֮һ����ش��������⣺
(1)��֪��N2(g)+O2(g)=2NO(g)��H1=+180.5kJ/mol
C(s)+O2(g)=CO2(g)��H2=-393.5kJ/mol
2C(s)+O2(g)=2CO(g)��H3=-221kJ/mol
��ij��Ӧ��ƽ�ⳣ������ʽΪ��K=[c(N2) ��c2(CO2)]/[c2(NO) ��c2(CO)],��˷�Ӧ���Ȼ�ѧ����ʽΪ____________________________________��
(2)N2O5��һ�������¿ɷ����ֽ⣺2N2O5(g)![]() 4NO2(g)+O2(g),ij�¶��º����ܱ������м���һ����N2O5,���N2O5Ũ����ʱ��ı仯���±���
4NO2(g)+O2(g),ij�¶��º����ܱ������м���һ����N2O5,���N2O5Ũ����ʱ��ı仯���±���
t/min | 0 | 1 | 2 | 3 | 4 | 5 |
c(N2O5)/<mol��L-1) | 1.00 | 0.71 | 0.50 | 0.35 | 0.25 | 0.17 |
�ٷ�Ӧ��ʼʱ��ϵѹǿΪp0,��2minʱ��ϵѹǿΪp1����p1��p0=__________��2~5min����NO2��ʾ�ĸ÷�Ӧ��ƽ����Ӧ����Ϊ__________��
��һ���¶��£��ں����ܱ������г���һ����N2O5���и÷�Ӧ,���жϷ�Ӧ�Ѵﵽ��ѧƽ��״̬����__________��(����ĸ)��
a��NO2��O2��Ũ�ȱȱ��ֲ��� b��������ѹǿ���ٱ仯
c��2v��(NO2)=v��(N2O5) d��������ܶȱ��ֲ���
(3)Kp���÷�Ӧ��ϵ���������ʵķ�ѹ����ʾ��ƽ�ⳣ��������K����ʽ��ƽ��Ũ����ƽ���ѹ���档��֪��Ӧ��NO2(g)+CO(g)![]() NO(g)+CO2(g),�÷�Ӧ������Ӧ����v��=k����p(NO2)��p(CO),�淴Ӧ����v��=k����p(NO)��p(CO2),����k����k��Ϊ���ʳ���,��KpΪ__________(��k����k����ʾ)��
NO(g)+CO2(g),�÷�Ӧ������Ӧ����v��=k����p(NO2)��p(CO),�淴Ӧ����v��=k����p(NO)��p(CO2),����k����k��Ϊ���ʳ���,��KpΪ__________(��k����k����ʾ)��
(4)��ͼ���ܱշ�Ӧ���а�n(N2) ��n(H2)=1��3Ͷ�Ϻ�,��200�桢400�桢600����,�ϳ�NH3��Ӧ�ﵽƽ��ʱ,�������NH3�����ʵ���������ѹǿ�ı仯���ߣ���֪�÷�ӦΪ���ȷ�Ӧ��
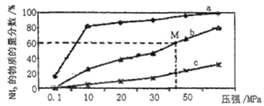
������a��Ӧ���¶���__________��
��M���Ӧ��H2��ת������__________��
(5)��ҵ�ϳ��ð�ˮ���ն�������,������(NH4)2SO3���жϳ�����(NH4)2SO3��Һ������Բ�˵���ж����ݣ�______________________________________________��(��֪��NH3��H2O��Kb=1.8��10-5;H2SO3��Ka1=1.3��10-2,Ka2=6.3��10-8��)