��Ŀ����
����Ŀ����ͼ��ʾAE�������ʼ���ת����ϵ������AΪ����ɫ���壬BΪ���ʡ����Ƴ������ʵĻ�ѧʽ����д��B��C��C��D��D��E��Ӧ�Ļ�ѧ����ʽ��
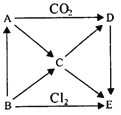
(1)д��B��C��C��D��D��E��Ӧ�Ļ�ѧ����ʽ��
B��C��_______________��
C��D��_______________��
D��E��_______________��
(2)��3.9 gAͶ�뵽������ˮ�г�ַ�Ӧ�����ɵ������ڱ�״���µ����Ϊ_____L������Ӧ�����Һ���1000mL��Һ��������Һ�����ʵı�Ũ��Ϊ______________��
���𰸡� 2Na��2H2O=2NaOH��H2�� 2NaOH��CO2=Na2CO3��H2O Na2CO3��2HCl=2NaCl��CO2����H2O 0.56 0.1 mol��L-1
��������B�ǵ��ʣ�AΪ����ɫ���壬A�ܺ�CO2��Ӧ����A��Na2O2��B�ܺ�������Ӧ����B��Na��E��NaCl��A�Ͷ�����̼��Ӧ����Na2CO3��O2��D��ת��ΪNaCl����D��Na2CO3��A��B��ת��ΪC��C��ת��ΪE��D����C��NaOH��
(1)B��Na������ˮ��Ӧ����NaOH����Ӧ��ѧ����ʽΪ2Na+2H2O�T2NaOH+H2�������������������̼��Ӧ��������̼���ƣ���Ӧ�Ļ�ѧ����ʽΪ2NaOH��CO2=Na2CO3��H2O��
D��Na2CO3���������ᷴӦ����NaCl����Ӧ�Ļ�ѧ����ʽΪNa2CO3+2HCl�T2NaCl+H2O+CO2�����ʴ�Ϊ��2Na+2H2O�T2NaOH+H2����2NaOH��CO2=Na2CO3��H2O��Na2CO3+2HCl�T2NaCl+H2O+CO2����
(2)n(Na2O2)=![]() =0.05mol��
=0.05mol��
�� 2Na2O2+2H2O=4NaOH+O2��
2mol 36g 4mol 22.4L
0.05mol m(H2O)n(NaOH) V(O2)
V(O2)=![]() =0.56L��n(NaOH)=
=0.56L��n(NaOH)=![]() =0.1mol��c(NaOH)=
=0.1mol��c(NaOH)=![]() =0.1mol/L���ʴ�Ϊ��0.56��0.1��
=0.1mol/L���ʴ�Ϊ��0.56��0.1��

 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�����Ŀ����1��ijС����������KMnO4��Һ��H2C2O4��Һ�ķ�Ӧ���˷�ӦΪ���ȷ�Ӧ����̽���������Ի�ѧ��Ӧ���ʵ�Ӱ�죬����������µķ�����¼ʵ������������Һ�������仯����
��ѡ�Լ���������0.20 mol/L H2C2O4��Һ��0.010 mol/L KMnO4��Һ�����ԣ���MnSO4������ˮ���Թܡ���Ͳ�����������ˮԡ�ۡ�
������ ��� | V��0.20 mol/L H2C2O4��Һ��/mL | V������ˮ��/mL | V��0.010 mol/L����KMnO4��Һ��/mL | m��MnSO4��/g | T/�� | �� |
�� | 2.0 | 0 | 4.0 | 0 | 50 | |
�� | 2.0 | 0 | 4.0 | 0 | 25 | |
�� | 1.5 | a | 4.0 | 0 | 25 | |
�� | 2.0 | 0 | 4.0 | 0.1 | 25 |
�ش��������⣺
��д��������Ӧ�����ӷ���ʽ��________________________��
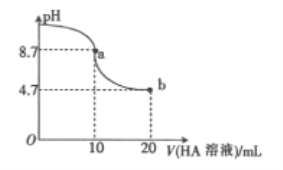
������ʵ��٢���̽��____________�Ի�ѧ��Ӧ���ʵ�Ӱ�죻����ʵ��ڢ���̽��____________�Ի�ѧ��Ӧ����
Ӱ�졣������ʵ��ڢ���̽��Ũ�ȶԻ�ѧ��Ӧ���ʵ�Ӱ�죬��aΪ______________�������е���������дt/s�����������______________________��
��ʵ����ж��������ظ��������Σ�������ֵ�ֱ�13.6��13.5��13.4�����Ի��ǰ����Һ�����С�仯�����ʱ����ƽ����Ӧ����v(KMnO4)��___________ ��
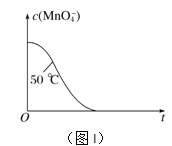
����֪ʵ���50��ʱc(MnO![]() )����Ӧʱ��t�ı仯������ͼI�������������������䣬��������ͼ�У�����ʵ���25��ʱc(MnO
)����Ӧʱ��t�ı仯������ͼI�������������������䣬��������ͼ�У�����ʵ���25��ʱc(MnO![]() )��t�ı仯����ʾ��ͼ��______________
)��t�ı仯����ʾ��ͼ��______________
 ����
����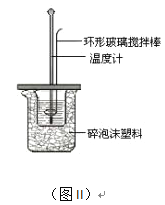
��2���ⶨ�к��ȵ�ʵ��װ����ͼII��ʾ��
��д��ϡ�����ϡ����������Һ��Ӧ��ʾ�к��ȵ��Ȼ�ѧ����ʽ_______________________���к�����ֵΪ57.3 kJ/mol����
��ȡ60mL0.50mol/L NaOH��Һ��50mL0.50mol/L������Һ����ʵ�飬ʵ���������±���
ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶� t2/�� | �¶Ȳ� ��t2-t1��/�� | ||
H2SO4 | NaOH | ƽ��ֵ | |||
1 | 26.2 | 26.0 | 26.1 | 29.9 | |
2 | 26.2 | 26.4 | 26.3 | 30.6 | |
3 | 25.9 | 25.9 | 25.9 | 29.5 | |
4 | 26.4 | 26.2 | 26.3 | 30.0 | |
������Ϊ0.50 mol/L NaOH��Һ��0.50 mol/L������Һ���ܶȶ���1 g/cm3���кͺ�������Һ�ı�����c=4.18 J/(g����)�����к�����H=________________________kJ/mol���г�����ʽ����
���������к���ƫ�ߵ�ԭ���ǣ�����ĸ��__________��
A��ʵ��װ�ñ��¡�����Ч����
B����ȡNaOH��Һ�����ʱ���Ӷ���
C����50mL0.50mol/L���������������з�Ӧ��
D�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶�
E. ��ȡ������Һ�����ʱ���Ӷ���